Astrazeneca Vaccine Phase 3
22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and. 25032021 AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety.
 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
02092020 On Monday the pharmaceutical company revealed its Covid-19 vaccine candidate entered the final testing phase in the United States a Phase 3 clinical trial.
Astrazeneca vaccine phase 3. COV003 is a continuing single-blind phase 3 study in Brazil that began on June 23 2020. AstraZeneca has begun a US. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US.
Phase 3 clinical trial of its COVID-19 vaccine candidate AZD1222. UPDATED ON OCT 24 2020 1246 PM IST The Brihanmumbai Municipal Corporation BMC has completed the second phase of clinical trials of the Oxford-AstraZeneca Covid-19. The focus of recruitment was targeted at those at high risk of exposure to the virus including health-care workers at six sites across Brazil.
Oxford Universitys professor Sarah Gilbert said that work to prepare the coronavirus vaccine for submission to the US. Once the vaccine moves into phase 3 the researchers will limit enrollment to people age 18 years and older. Actual Primary Completion Date.
19012021 Moreover the current phase 3 trials are powered to detect protection against symptomatic infections. The company now says. Actual Study Start Date.
13042021 Vaccines in Phase 3 Clinical Trials As of February 27 2021 large-scale Phase 3 clinical trials are in progress or being planned for two COVID-19 vaccines in the United States. However protection against severe. 22032021 What happens now AstraZenecas Covid vaccine has completed Phase 3 trial.
10 10 viral particles per dose administered 28 days apart but a subset LDSD cohort in one of the UK trials inadvertently received a half-dose of the vaccine low dose as the first dose before a change in. The BARDA-funded study will enroll 30000 adults at sites in the US. AstraZeneca COVID-19 vaccine Novavax COVID-19 vaccine.
Estimated Study Completion Date. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. 20072020 According to AstraZeneca late-stage trials of AZD1222 include a Phase III trial in Brazil 2000 participants ISRCTN89951424 a Phase IIbIII trial in the UK.
31082020 CNN British drugmaker AstraZeneca said Monday it has started Phase 3 trials of its experimental coronavirus vaccine in the United States. And some other countries in. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704.
With a 76 efficacy rate against symptomatic COVID. Adult participants in the phase 2 and 3 trials will be randomized to receive one or two. After phase 1 results supported a two-dose regimen the trial protocols were amended where necessary to require two standard doses SDSD cohort of approximately 5.
Co-invented by scientists at the. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
 Covid 19 Vaccine These Countries Have Started Rolling Out Coronavirus Vaccine Check Full List Here
Covid 19 Vaccine These Countries Have Started Rolling Out Coronavirus Vaccine Check Full List Here
 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
 Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
 Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India
Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India

 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Astrazeneca Says Shot 90 Effective
Astrazeneca Says Shot 90 Effective
 Oxford Astrazeneca Covid Vaccine Prompts Immune Response In All Adults
Oxford Astrazeneca Covid Vaccine Prompts Immune Response In All Adults
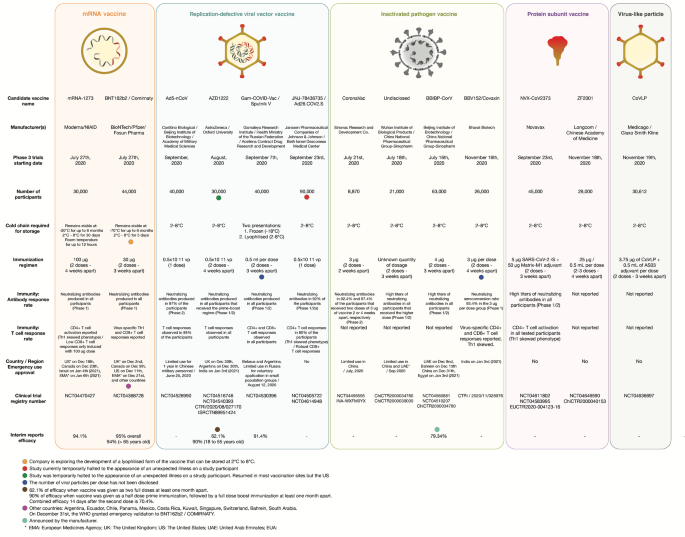
 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines

Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post
Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post
 Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
 5 Things You Need To Know About The Astrazeneca Vaccine Now The Tga Has Approved It For Use In Australia Rmit University
5 Things You Need To Know About The Astrazeneca Vaccine Now The Tga Has Approved It For Use In Australia Rmit University
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
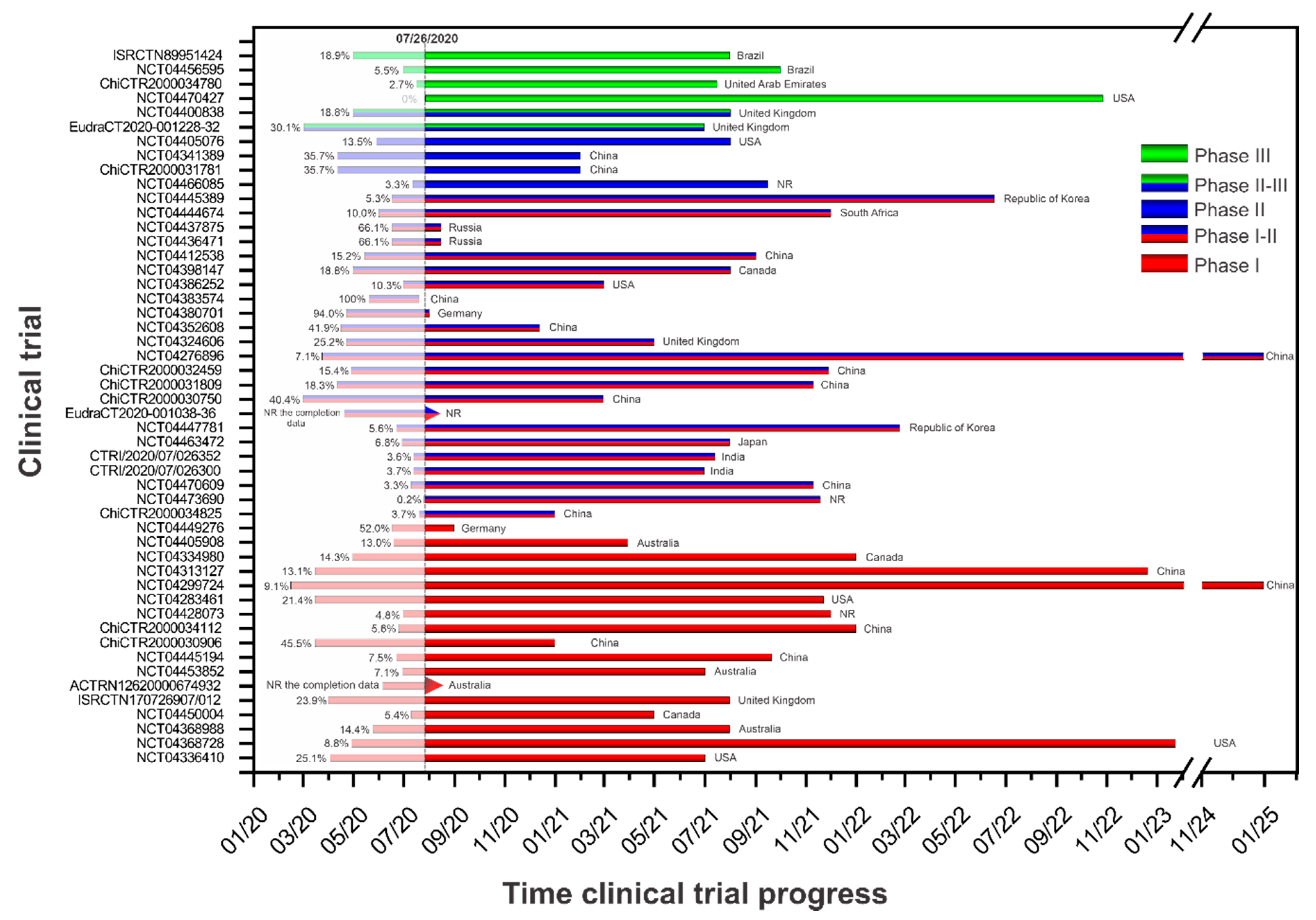 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Post a Comment for "Astrazeneca Vaccine Phase 3"