Astrazeneca Vaccine Phase 3 Result
Actual Study Start Date. On 22 March 2021 AstraZeneca released interim results from the phase III trial conducted in the US that showed efficacy of 79 at preventing symptomatic COVID-19 and 100 efficacy at preventing severe disease and hospitalisation.
 Explained Here Are The Key Takeaways From Oxford S Covid 19 Vaccine Error Explained News The Indian Express
Explained Here Are The Key Takeaways From Oxford S Covid 19 Vaccine Error Explained News The Indian Express
16112020 Interim results from phase 3 clinical trials of the Covid vaccine from US company Moderna has revealed it to be have almost 95 efficacy at preventing the disease.

Astrazeneca vaccine phase 3 result. New vaccine efficacy results are reported now in The Lancet. Results demonstrated vaccine efficacy of 76 CI. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19.
Another vaccine option in coming months. Results from the subgroup comparisons presented in this analysis were similar to overall results table 3. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older.
Tuesdays results the first Phase 3 clinical data for a. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19. AstraZeneca and the University of Oxford fail to impress the markets with phase 3 trial results.
08032021 While the vaccine appears to meet the FDAs standards for authorization its future in the US. 08122020 Topline data on the AstraZeneca vaccine from a Phase 3 trial were released in November. Had bet more on AstraZenecas vaccine than any other and even with three alternatives available could still benefit from its availability.
22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and. Oxford and AstraZeneca researchers present a pooled analysis in The Lancet of Phase 3 trials of a coronavirus vaccine resulting in an average efficacy of 704. 03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose.
AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US. Modernas phase 3 analysis involved 30000 people in the US half given the vaccine and. At one point the US.
In the SDSD UK cohort who were aged 1855 years 49 cases were available for inclusion in the analysis and vaccine efficacy was 593 95 CI 251 to 779. 30112020 The UK has bought 100m doses of the Oxford vaccine which will be a core part of its mass immunisation plans. First peer-reviewed results of phase 3 human trials of Oxford coronavirus vaccine demonstrate efficacy University of Oxford.
Study results showed that a single dose of the vaccine led to a four-fold increase in antibodies to the SARS-CoV-2 spike protein in 95 of participants one month following. Findings were published in The Lancet journal. 59 to 86 after a first.
Hinges on forthcoming results from a large Phase 3 study run in the country and in South America. Actual Primary Completion Date. But in order to do so the company needs to.
Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation. The news followed an announcement. Logistics and production could favor their vaccine however.
20072020 These results together with the induction of both humoral and cellular immune responses support large-scale evaluation of this candidate vaccine in an ongoing phase 3 program the researchers. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun.
20042021 In the Phase III COV001 trial conducted in the UK the vaccine was tolerated and generated strong immune responses in all evaluated participants. Estimated Study Completion Date.
 Astrazeneca Oxford S Covid 19 Vaccine Could See Increased Global Uptake
Astrazeneca Oxford S Covid 19 Vaccine Could See Increased Global Uptake
 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
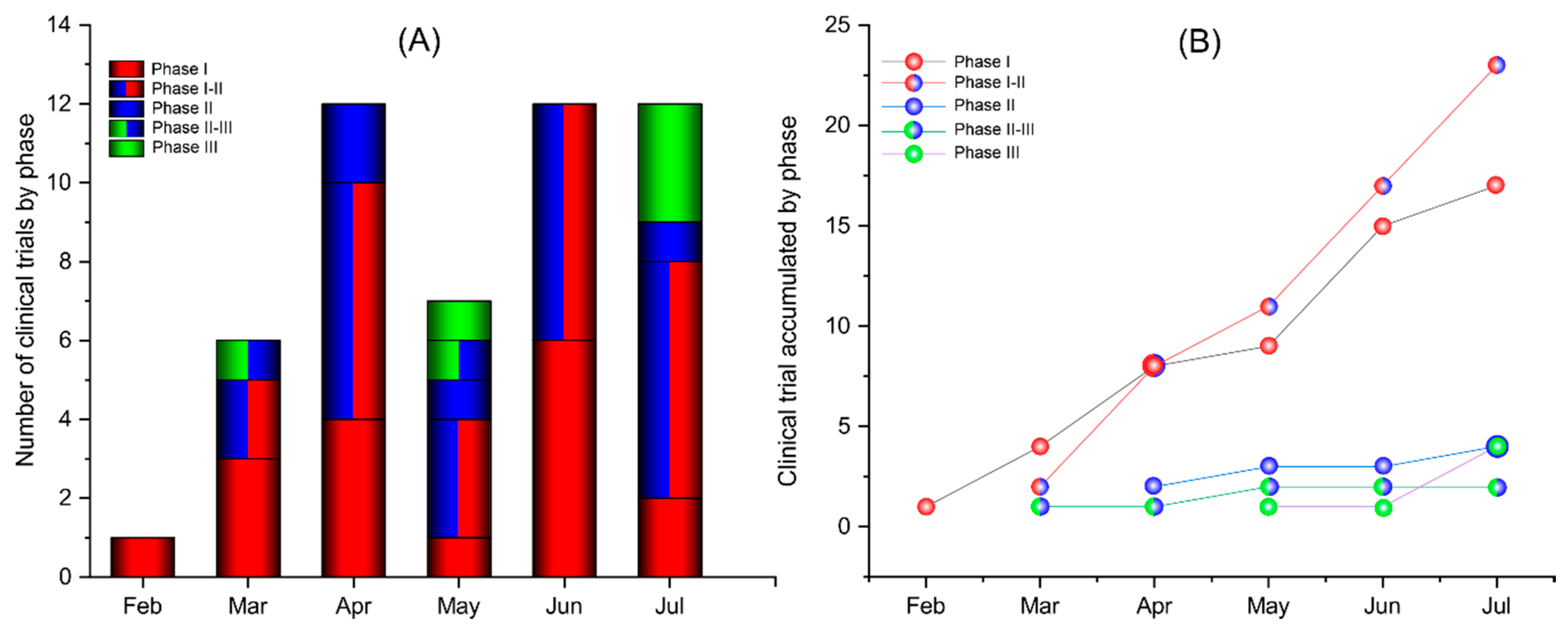 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
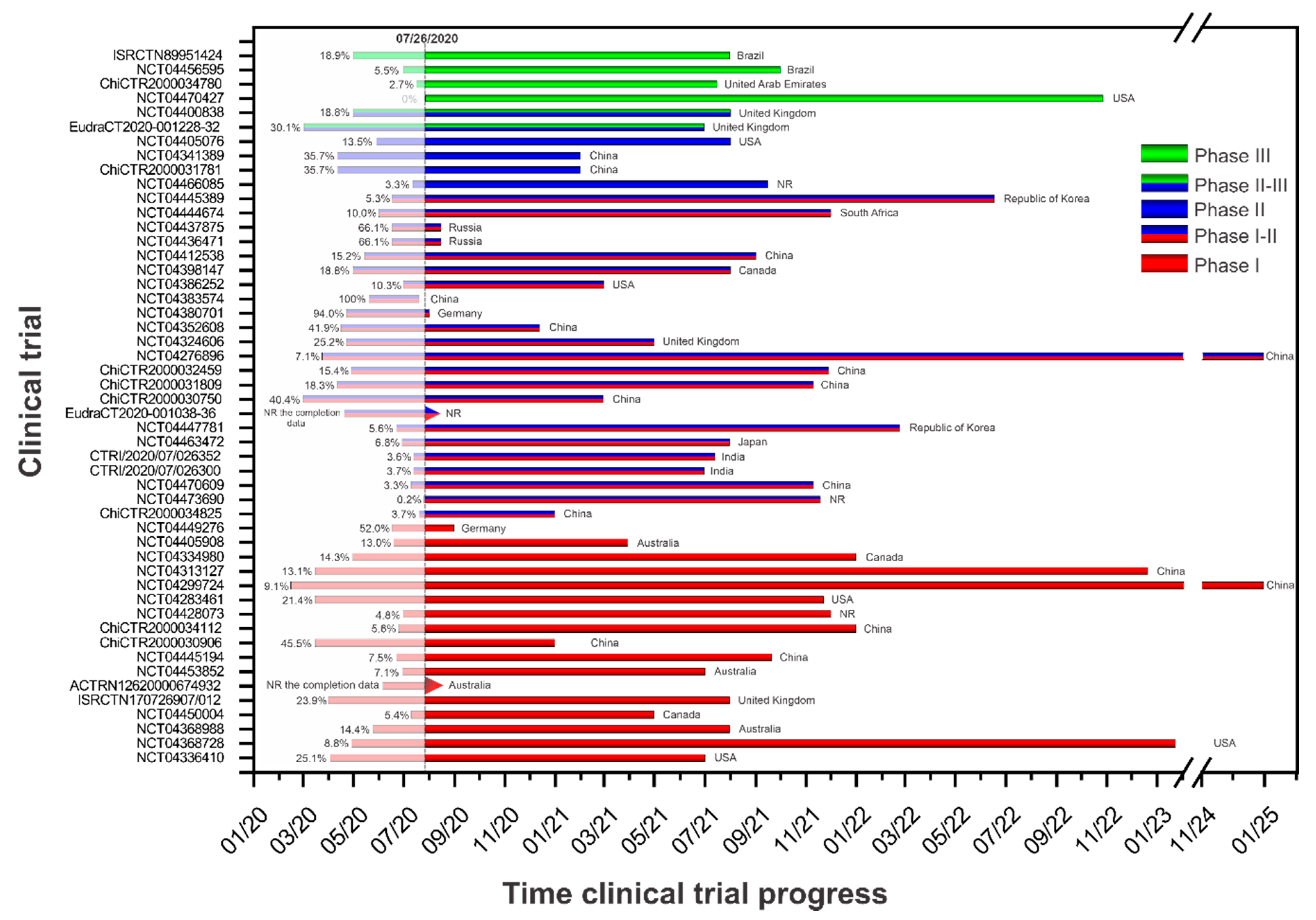 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
 Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
 Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
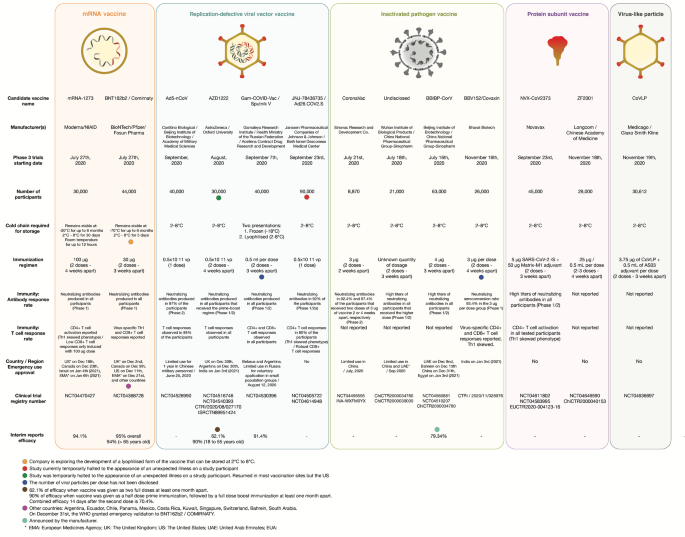 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
 Astrazeneca To Give 400m Covid 19 Vaccine Doses To Europe Data Expected August September 2020 06 14 Bioworld
Astrazeneca To Give 400m Covid 19 Vaccine Doses To Europe Data Expected August September 2020 06 14 Bioworld
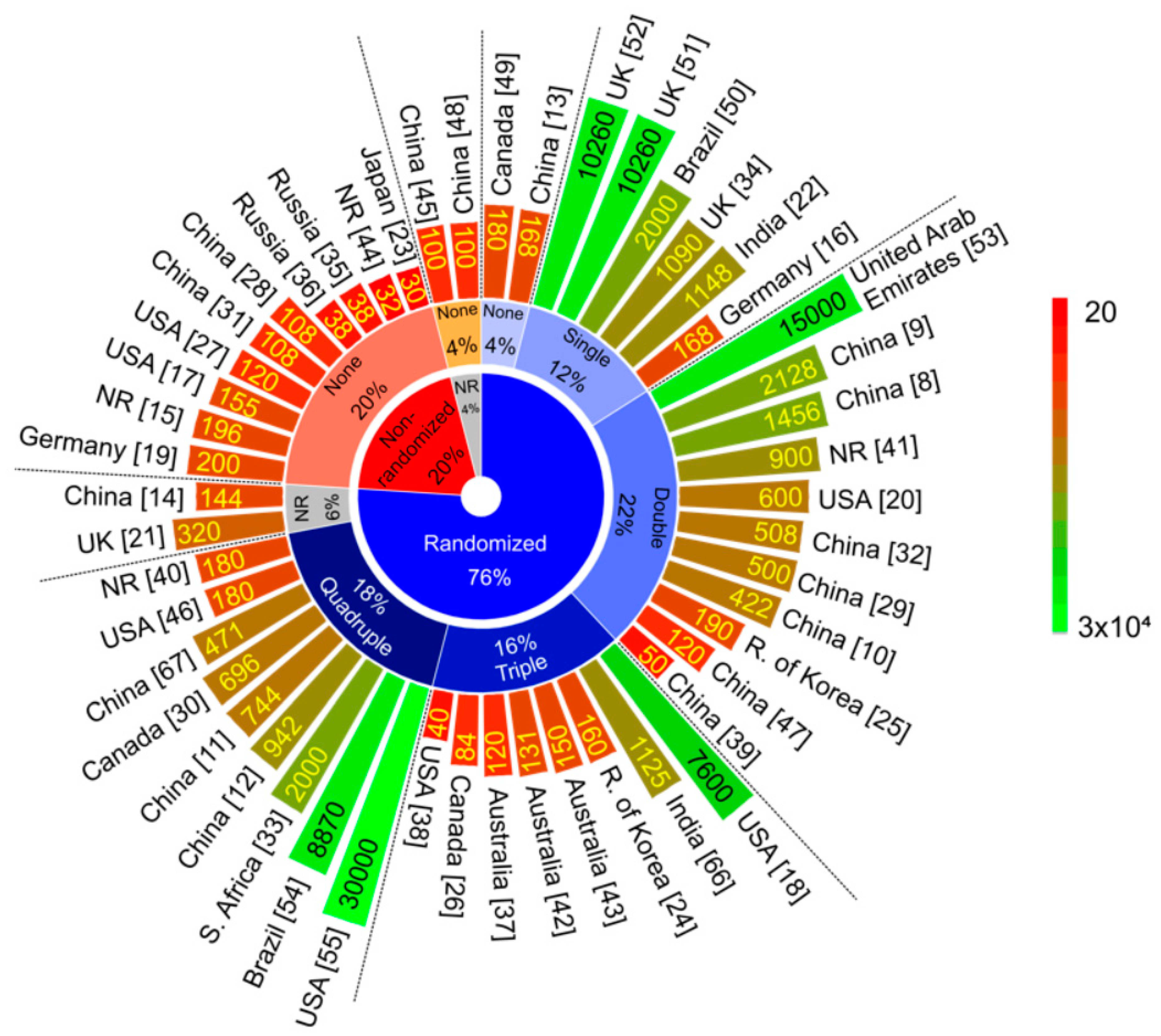 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html

Post a Comment for "Astrazeneca Vaccine Phase 3 Result"