Astrazeneca Phase 3 Trial Results
Results demonstrated vaccine efficacy of 76 CI. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older.
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation.

Astrazeneca phase 3 trial results. AZNL AZN announced Monday positive high-level results from the ELEVATE-RR Phase III trial of Calquence against ibrutinib in chronic lymphocytic. We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
The results of the phase 3 clinical trials of the Oxford AstraZeneca COVID-19 vaccine candidate have been published in The Lancet. 25012021 RTTNews - British drug major AstraZeneca Plc. 22032021 The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79 at preventing symptomatic COVID-19 and 100 efficacy at preventing severe disease and hospitalisation.
A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US. Partnered with one of the worlds leading contract research organizations CROs called IQVIA AstraZeneca has launched the Phase 3 double-blind placebo-controlled study of AZD1222 for the possible prevention of COVID-19 in adults.
These results have been presented to the independent Data Safety Monitoring Board. The interim analysis for efficacy was based on. 25032021 Positive high-level results from the primary analysis of the Phase III trial of AZD1222 in the US have confirmed vaccine efficacy consistent with the pre-specified interim analysis announced on Monday 22 March 2021.
Because we operate in an ever-changing environment. This web site provides clinical trial data results and other information from or regarding AstraZeneca-sponsored clinical trials. AZD 1222 Phase 3 Clinical Trial Comes to America This study literally just started last week according to US.
13012020 the trial as Epanova is unlikely to demonstrate a benefit to patients Following the recommendation from an independent Data Monitoring Committee AstraZeneca has decided to close the Phase III STRENGTH trial for Epanova omega-3 carboxylic acids due to its low likelihood of demonstrating a benefit to patients with mixed dyslipidaemia MDL who are at increased risk. The company now says. They both started phase 3 trials in the US on the same day at the end of July although the BNTPfizer one is a compressed phase 123 trial.
20072020 According to AstraZeneca late-stage trials of AZD1222 include a Phase III trial in Brazil 2000 participants ISRCTN89951424 a Phase IIbIII trial in. New vaccine efficacy results are reported now in The Lancet. This site is part of our commitment to provide patients and healthcare professionals with meaningful information about AstraZeneca medicines and drugs in development.
The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019. 11092020 Overview of phase 3 trials with earlier phase results The first 5 trials likely to have phase 3 results are rounded out by Germanys BNTPfizer vaccine and the NIHModerna vaccine from the US. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector.
Oxford and AstraZeneca researchers present a pooled analysis in The Lancet of Phase 3 trials of a coronavirus vaccine resulting in an average efficacy of 704. Expert reaction to a study on the results of phase 3 human trials of the OxfordAZ COVID-19 vaccine. This Roundup accompanied an SMC Briefing.
03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose. This interim safety and efficacy analysis was based on 32449 participants accruing 141 symptomatic cases of COVID-19. First peer-reviewed results of phase 3 human trials of Oxford coronavirus vaccine demonstrate efficacy University of Oxford.
 The 111 Study A Single Arm Phase 3 Trial Evaluating One Cycle Of Bleomycin Etoposide And Cisplatin As Adjuvant Chemotherapy In High Risk Stage 1 Nonseminomatous Or Combined Germ Cell Tumours Of The Testis
The 111 Study A Single Arm Phase 3 Trial Evaluating One Cycle Of Bleomycin Etoposide And Cisplatin As Adjuvant Chemotherapy In High Risk Stage 1 Nonseminomatous Or Combined Germ Cell Tumours Of The Testis
Imfinzi Plus Tremelimumab Demonstrated Promising Clinical Activity And Tolerability In Patients With Advanced Liver Cancer
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
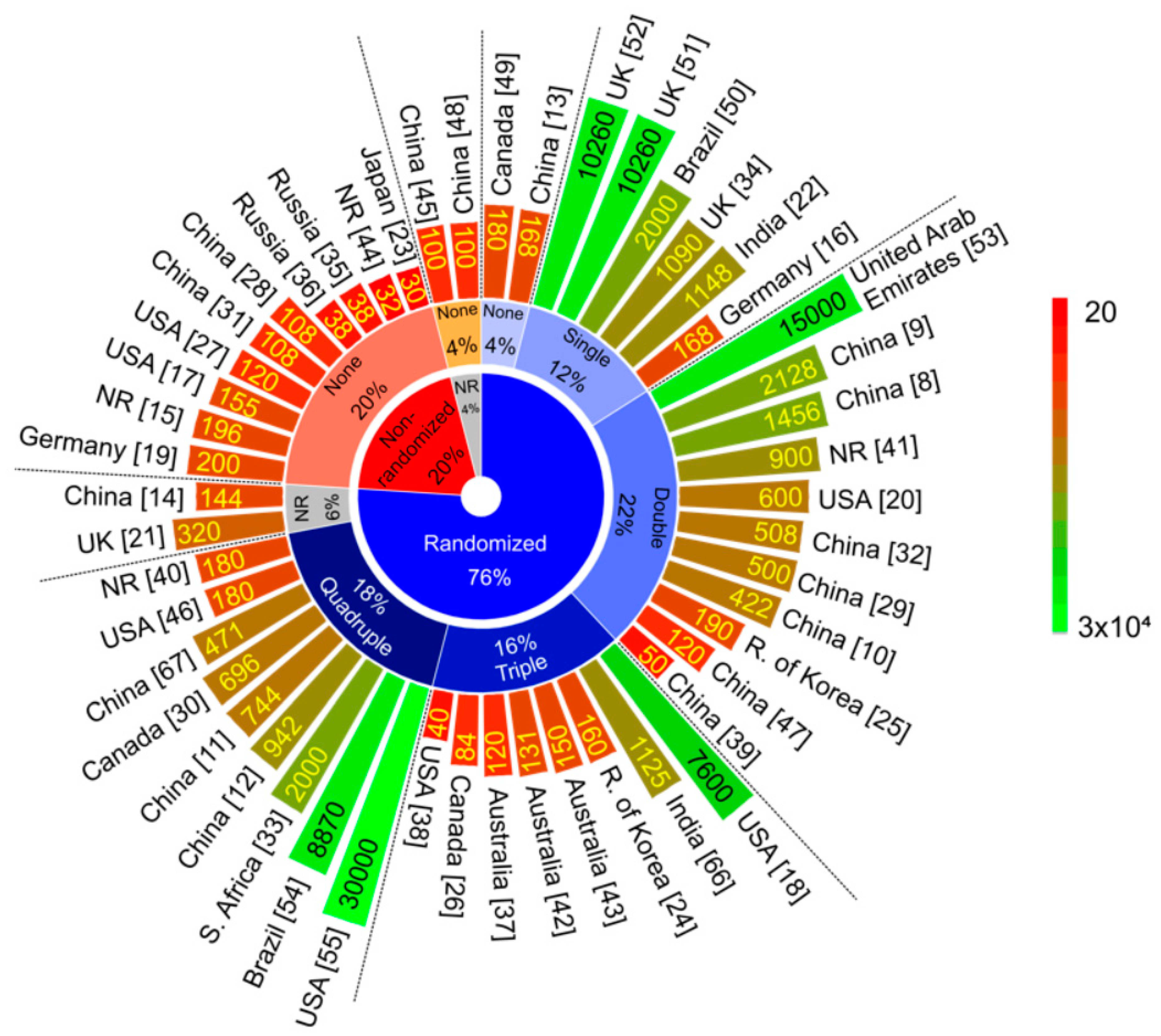 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Supplemental Materials For Cediranib In Patients With Relapsed Platinum Sensitive Ovarian Cancer Icon6 A Randomised Double Blind Placebo Controlled Phase 3 Trial The Lancet
Supplemental Materials For Cediranib In Patients With Relapsed Platinum Sensitive Ovarian Cancer Icon6 A Randomised Double Blind Placebo Controlled Phase 3 Trial The Lancet
 Covid 19 Vaccines November Update Progress Of Clinical Trials Post
Covid 19 Vaccines November Update Progress Of Clinical Trials Post

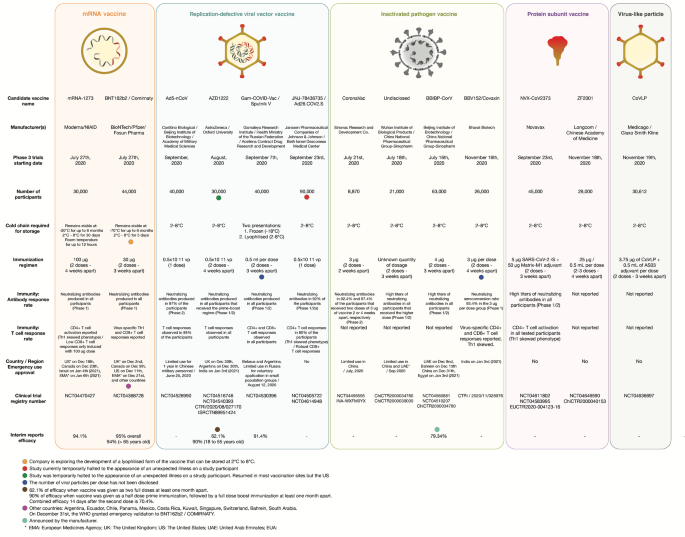 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
 Astrazeneca Merck Co S Lynparza Gets Eu Approval In Prostate Cancer S P Global Market Intelligence
Astrazeneca Merck Co S Lynparza Gets Eu Approval In Prostate Cancer S P Global Market Intelligence
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
 Covid Vaccine Astrazeneca Phase Iii Trials Interim Results Youtube
Covid Vaccine Astrazeneca Phase Iii Trials Interim Results Youtube
 Effect Of Duration Of Adjuvant Chemotherapy For Patients With Stage Iii Colon Cancer Idea Collaboration Final Results From A Prospective Pooled Analysis Of Six Randomised Phase 3 Trials The Lancet Oncology
Effect Of Duration Of Adjuvant Chemotherapy For Patients With Stage Iii Colon Cancer Idea Collaboration Final Results From A Prospective Pooled Analysis Of Six Randomised Phase 3 Trials The Lancet Oncology
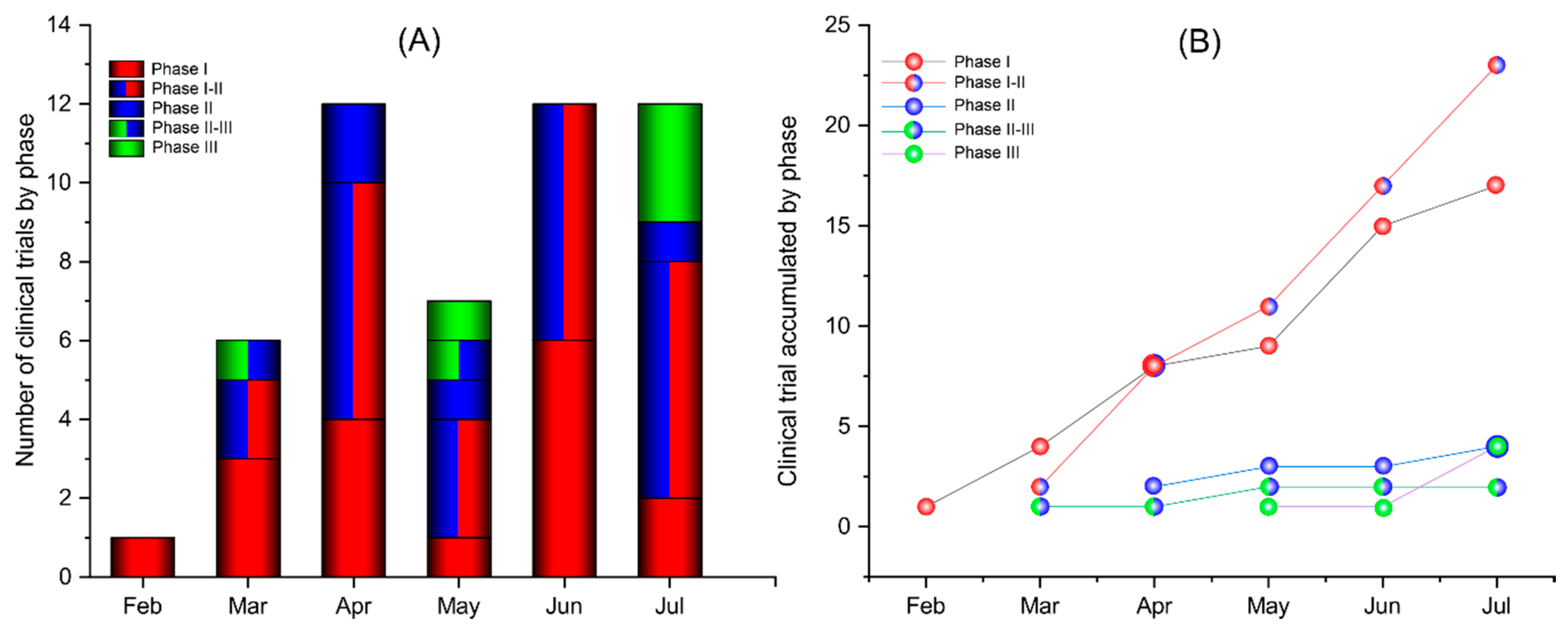 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Covid 19 Vaccines Set To Enter Phase Iii Trials In India Initial Results Highly Encouraging The Weather Channel Articles From The Weather Channel Weather Com
Covid 19 Vaccines Set To Enter Phase Iii Trials In India Initial Results Highly Encouraging The Weather Channel Articles From The Weather Channel Weather Com
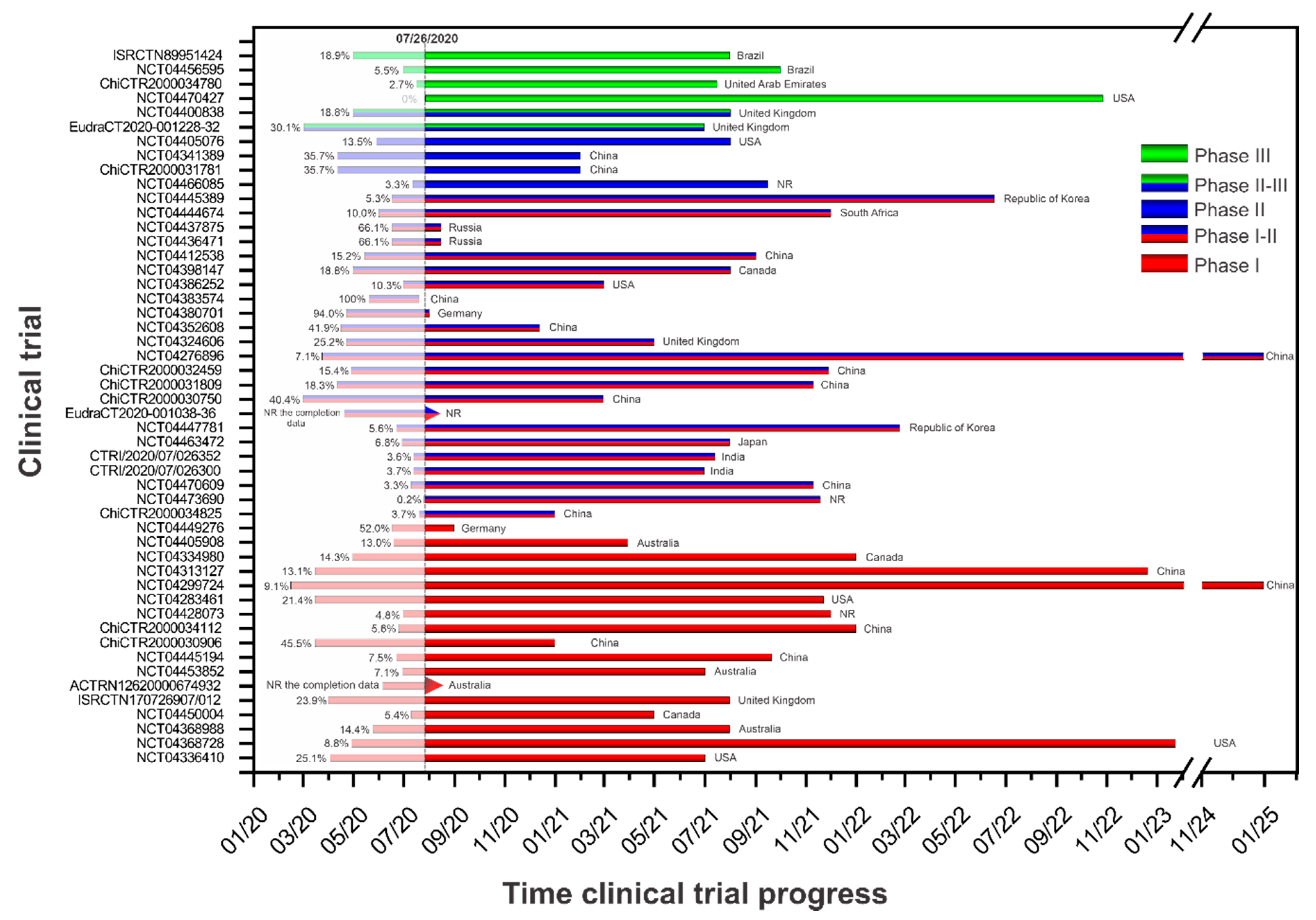 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Olaparib Tablets As Maintenance Therapy In Patients With Platinum Sensitive Relapsed Ovarian Cancer And A Brca1 2 Mutation Solo2 Engot Ov21 A Double Blind Randomised Placebo Controlled Phase 3 Trial The Lancet Oncology
Olaparib Tablets As Maintenance Therapy In Patients With Platinum Sensitive Relapsed Ovarian Cancer And A Brca1 2 Mutation Solo2 Engot Ov21 A Double Blind Randomised Placebo Controlled Phase 3 Trial The Lancet Oncology
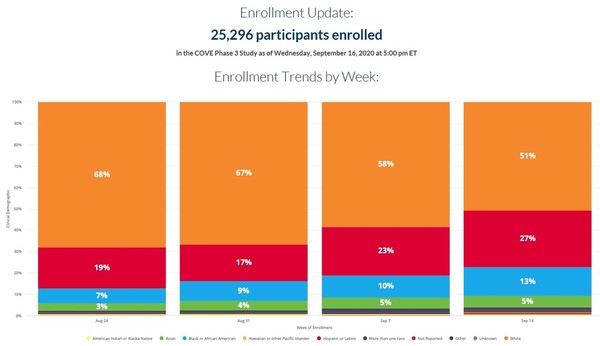
Post a Comment for "Astrazeneca Phase 3 Trial Results"