Astrazeneca Phase Iii Trial Covid
03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose. 12042021 Apr 12 2021 223AM EDT RTTNews - British drug major AstraZeneca Plc AZN AZNL Monday announced that its DARE-19 Phase III trial for Farxiga dapagliflozin in COVID-19 did not achieve.
 Astrazeneca S Covid 19 Vaccine Proves Safe And Effective In Phase Iii Trial
Astrazeneca S Covid 19 Vaccine Proves Safe And Effective In Phase Iii Trial
Actual Study Start Date.
Astrazeneca phase iii trial covid. An interim analysis of four randomised controlled trials in Brazil South Africa and the UK - The Lancet. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine AZD1222 against SARS-CoV-2. SeventyFour iStock The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79 at preventing symptomatic COVID-19 and 100 efficacy at preventing severe disease and hospitalisation. 26012021 Care Access Research is providing decentralized mobile research site operations for AstraZenecas AZD7442 phase III clinical trial in the US. 04022021 AstraZenecas Covid-19 vaccine proves safe and effective in Phase III trial.
AstraZeneca resumes US phase III COVID-19 vaccine trial. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. AstraZenecas COVID-19 vaccine has just undergone a phase III trial in the US demonstrating 79 efficacy at preventing symptomatic COVID-19 and 11 efficacy at preventing severe disease and hospitalization.
01092020 AstraZeneca said today it has launched a 30000-patient Phase III trial in the US. 12042021 Kezia Parkins 12th April 2021 Last Updated April 12th 2021 1552 Results from a Phase III clinical trial to see if AstraZenecas AZs diabetes and heart failure drug Farxiga dapagliflozin could help treat high-risk patients hospitalised with Covid-19 have shown that the drug failed to make an impact. 27th October 2020.
22032021 Results of the trial which involved more than 32000 volunteers showed two doses of the vaccine administered four weeks apart had an efficacy of 79 at preventing symptoms of. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19.
This interim safety and efficacy analysis was based on 32449 participants accruing 141 symptomatic cases of COVID-19. Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704. Estimated Primary Completion Date.
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine. 22032021 March 22 2021 0700 AM Eastern Daylight Time WILMINGTON Del-- BUSINESS WIRE--The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79 at. 22032021 LONDONTAIPEI AstraZenecas COVID-19 vaccine performed better than expected in a major late-stage trial potentially paving the way.
AstraZeneca has resumed all clinical trials of its coronavirus vaccine candidate after the Food and Drug Administration FDA authorised the restart in the US. 04 Feb 2021 Last Updated February 4th 2021 1506 AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing. Vaccine efficacy was consistent across ethnicity and.
ChAdOx1 nCoV-19 has an acceptable safety profile and has been found to be efficacious against symptomatic COVID-19 in this interim analysis of ongoing clinical trials. Of AZD1222 the COVID-19 vaccine it is co-developing with the University of Oxford and a spinouta study that. 12042021 LONDON UK I April 12 2021 I AstraZeneca and Saint Lukes Mid America Heart Institute today announced high-level results of the primary analysis from the DARE-19 Phase III trial assessing the potential of Farxiga dapagliflozin to treat patients hospitalised with COVID-19 who are at risk of developing serious complications.
A Phase III Randomized Double-blind Placebo-controlled Multi-center Study in Adults to Determine the Safety and Efficacy of AZD7442 a Combination Product of Two Monoclonal Antibodies AZD8895 and AZD1061 for Pre-exposure Prophylaxis of COVID-19.

 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
 Astrazeneca Starts U S Final Stage Trial Of Covid 19 Vaccine Tif
Astrazeneca Starts U S Final Stage Trial Of Covid 19 Vaccine Tif
 Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
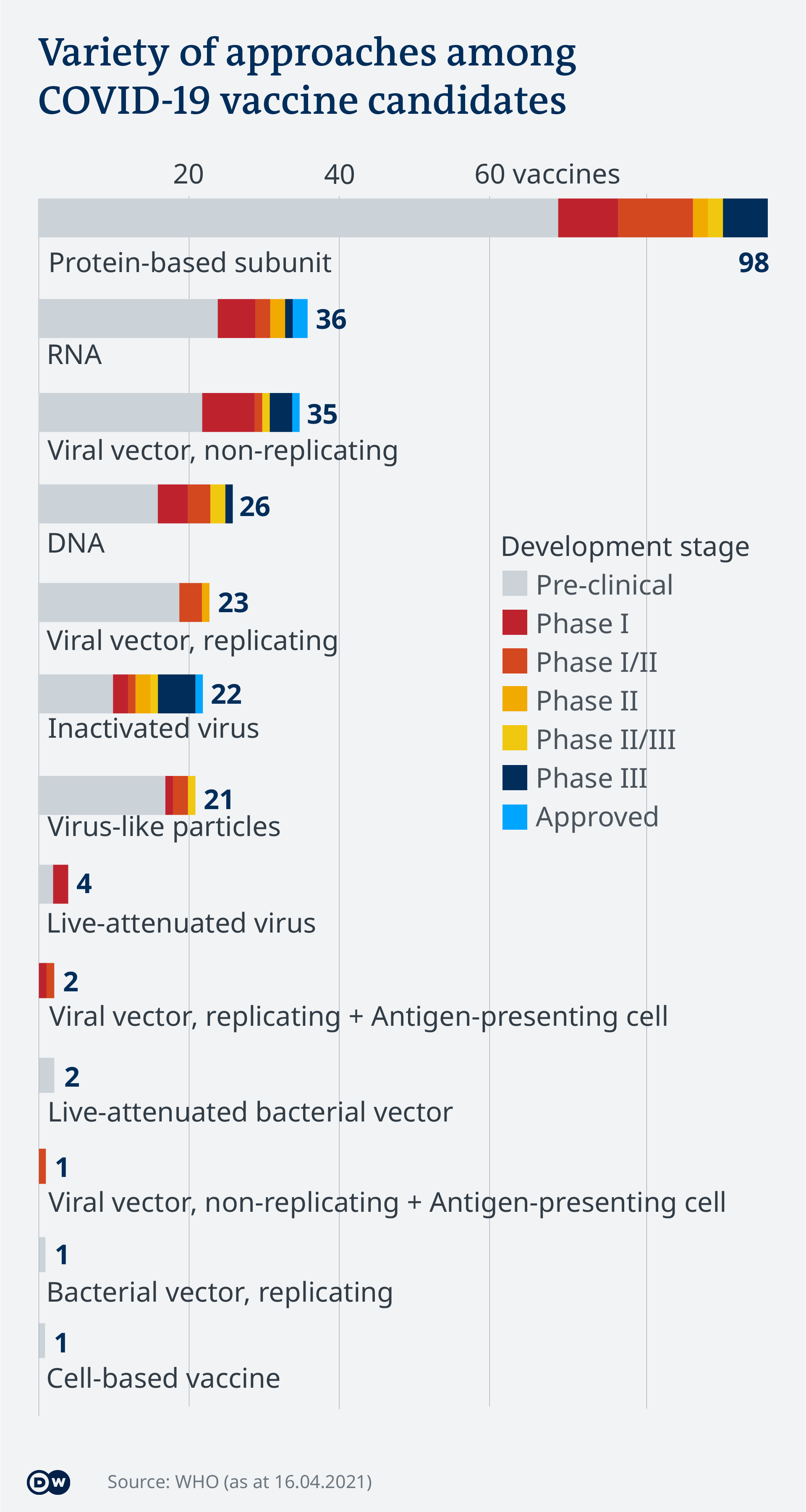
 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Astrazeneca Dagli Stati Uniti Conferme Dell Efficacia 100 Per Le Forme Gravi Di Covid Nessun Problema Di Trombosi Il Riformista
Astrazeneca Dagli Stati Uniti Conferme Dell Efficacia 100 Per Le Forme Gravi Di Covid Nessun Problema Di Trombosi Il Riformista
 Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India
Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India
 Astrazeneca S Covid 19 Vaccine Candidate Begins Phase 3 Trial Rt
Astrazeneca S Covid 19 Vaccine Candidate Begins Phase 3 Trial Rt
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
 Astrazeneca Covid 19 Vaccine Completes Phase 3 Trial Showing 79 Percent Effectiveness
Astrazeneca Covid 19 Vaccine Completes Phase 3 Trial Showing 79 Percent Effectiveness
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Dare 19 Phase Iii Trial For Farxiga In Covid 19 Misses Endpoint
Dare 19 Phase Iii Trial For Farxiga In Covid 19 Misses Endpoint
 Denis Wirtz On Twitter There Are Now 8 Covid 19 Vaccines Currently In Phase Iii Of Clinical Trials Including 1 Moderna Nih 2 Pfizer Biontech 3 Astrazeneca Oxford U 4 Murdoch Research Institute 5 Sinovac 6
Denis Wirtz On Twitter There Are Now 8 Covid 19 Vaccines Currently In Phase Iii Of Clinical Trials Including 1 Moderna Nih 2 Pfizer Biontech 3 Astrazeneca Oxford U 4 Murdoch Research Institute 5 Sinovac 6

Post a Comment for "Astrazeneca Phase Iii Trial Covid"