Astrazeneca Covid Vaccine Phase 3 Results
The only Grade 3 severe solicited adverse events greater than or equal to 2 in frequency after the first or second dose was fatigue at 38 and headache at 20 following dose 2. But the company noted results were much better 90 for participants who had mistakenly received a half then full dose of vaccine rather than two full doses 62.
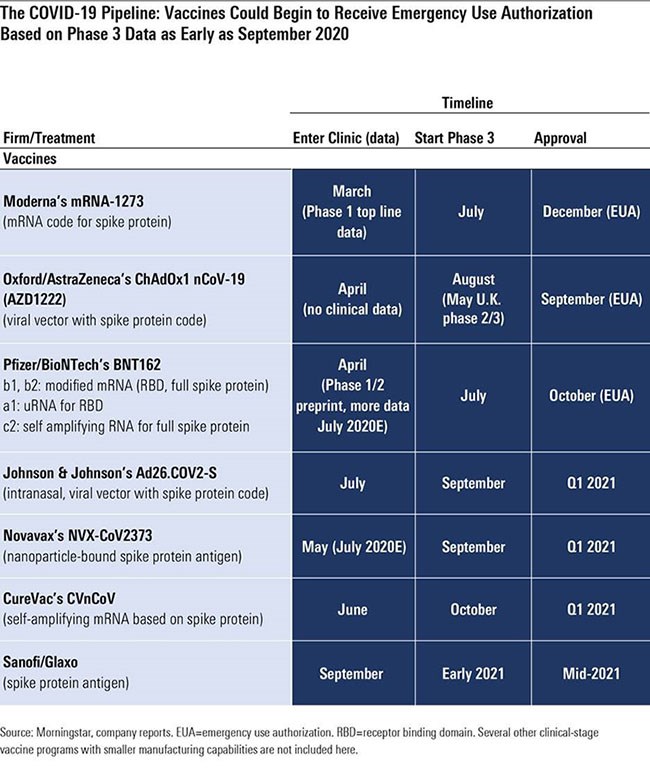 What A Covid 19 Vaccine Means For Investors Morningstar
What A Covid 19 Vaccine Means For Investors Morningstar
04022021 04 Feb 2021 Last Updated February 4th 2021 1506 AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing Covid-19 more than 22 days after the first dose.
Astrazeneca covid vaccine phase 3 results. An analysis published on 19 February 2021 showed an efficacy of 760 at preventing symptomatic COVID-19 beginning at 22 days following the first dose increasing to 813 when. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19. 22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and.
Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704. 25032021 AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety. Oxford and AstraZeneca researchers present a pooled analysis in The Lancet of Phase 3 trials of a coronavirus vaccine resulting in an average efficacy of 704.
The interim analysis for efficacy was based on 11636 participants accruing 131. Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation. Adult participants in the phase 2 and 3 trials will be randomized to receive one or two doses of AZD1222 or a vaccine against meningococcal bacteria that will serve as the control.
AstraZeneca COVID-19 vaccine phase 3 results show promise in elderly. Trial found its vaccine 79 effective but faced backlash after NIAID said Tuesday it may have included information that. 20072020 According to AstraZeneca late-stage trials of AZD1222 include a Phase III trial in Brazil 2000 participants ISRCTN89951424 a Phase IIbIII trial in.
A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. 19112020 AstraZeneca and Oxford University researchers expect to release full late-stage trial results by Christmas. 05112020 Pharmaceutical company AstraZeneca said Wednesday that phase three trial results for its COVID-19 vaccine could be ready later this year.
The company reported on Monday that a US. 08032021 When AstraZeneca first reported data in November it said a two-shot regimen was on average 70 effective at preventing COVID-19. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older.
Researchers show overall vaccine efficacy of 704 from a pooled analysis of two-dose regimen. The Oxford-AstraZeneca COVID19 vaccine is used to provide protection against infection by the SARS-CoV-2 virus in order to prevent COVID-19 in adults aged 18 years and older. With a 76 efficacy rate against symptomatic COVIDFlashback.
The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US. 19112020 Pharmaceutical company AstraZeneca on Thursday released data showing positive results from its Phase II COVID-19 vaccine trial particularly among adults over 56 years old.
We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa. Consistent with earlier shared results older adults tended to report fewer and milder solicited adverse events following vaccination. First peer-reviewed results of phase 3 human trials of Oxford coronavirus vaccine demonstrate efficacy University of Oxford.
New vaccine efficacy results are reported now in The Lancet.
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
 Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News

 Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
 Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
 Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
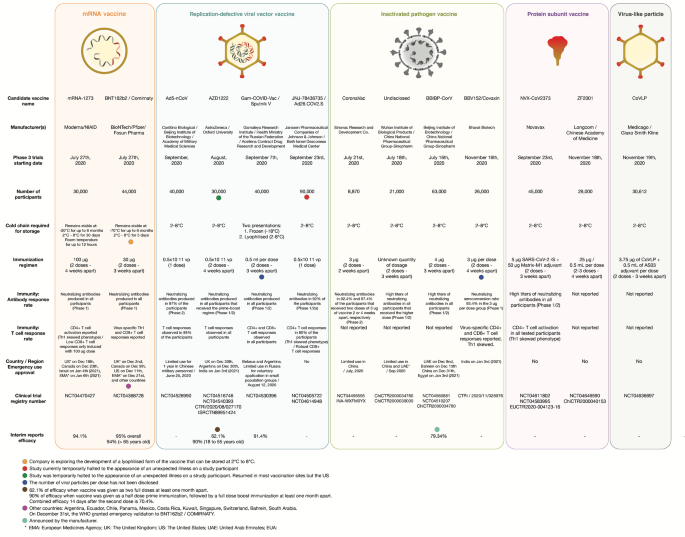 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
 Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa

 Astrazeneca Oxford S Covid 19 Vaccine Could See Increased Global Uptake
Astrazeneca Oxford S Covid 19 Vaccine Could See Increased Global Uptake
 How Soon Can We Expect Coronavirus Vaccine Crucial Data Expected By October End
How Soon Can We Expect Coronavirus Vaccine Crucial Data Expected By October End
Post a Comment for "Astrazeneca Covid Vaccine Phase 3 Results"