Astrazeneca Phase 3 Study
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. Actual Primary Completion Date.
 Adavosertib Plus Gemcitabine For Platinum Resistant Or Platinum Refractory Recurrent Ovarian Cancer A Double Blind Randomised Placebo Controlled Phase 2 Trial The Lancet
Adavosertib Plus Gemcitabine For Platinum Resistant Or Platinum Refractory Recurrent Ovarian Cancer A Double Blind Randomised Placebo Controlled Phase 2 Trial The Lancet
A Phase III Open-label Study in Adults to Determine the Safety and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19 Actual Study Start Date.

Astrazeneca phase 3 study. 22032021 The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79 at preventing symptomatic COVID-19 and 100 efficacy at preventing severe disease and hospitalisation. AstraZeneca After facing criticism over a preliminary report earlier this week we published updated Phase 3 study data for the Covid-19 vaccine on Wednesday. Results demonstrated vaccine efficacy of 76 CI.
22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and. Phase 3 Clinical Testing in the US. The company Currently the vaccine is said to be 76 effective In protecting from symptomatological cases of the virus.
Actual Study Start Date. A release published on Monday reported a 79 symptom efficacy rate. Estimated Study Completion Date.
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. This interim safety and efficacy analysis was based on 32449 participants accruing 141 symptomatic cases of COVID-19. 23032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy against symptomatic COVID-19 80 efficacy in people aged 65 years and older and 100 efficacy against severe or critical disease and hospitalization.
03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose. 23032021 In the Phase 3 study involving more than 32000 people AstraZeneca found that its vaccine was 79 efficacious in protecting against symptoms of COVID-19. Although there are no licensed vaccines against COVID-19 48 potential vaccine candidates based on a variety of platforms including lipid nanoparticle mRNA DNA adjuvanted protein inactivated virus particles and non-replicating viral vectors are in clinical trials of which 11 candidates are in phase 3 trials and a further 164 candidates are in preclinical testing.
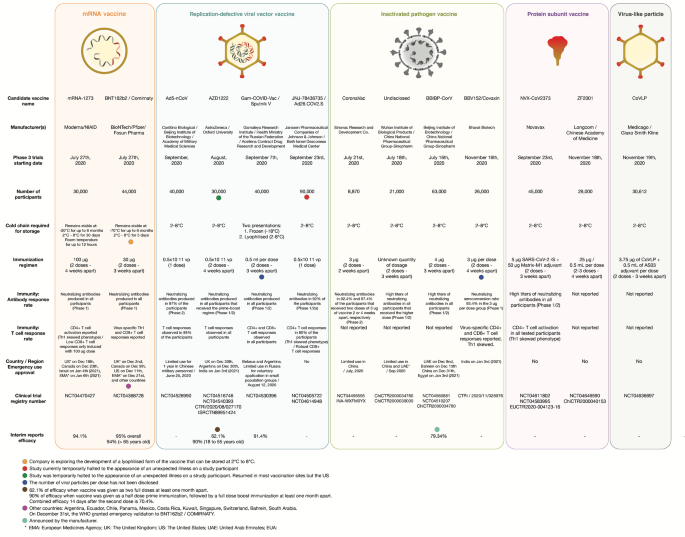 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
Imfinzi Plus Tremelimumab Demonstrated Promising Clinical Activity And Tolerability In Patients With Advanced Liver Cancer
 The Astrazeneca Phase 3 Covid 19 Vaccine Study Unprecedented Compression Of Timeline Of Est Start Date To Est Primary Completion Date
The Astrazeneca Phase 3 Covid 19 Vaccine Study Unprecedented Compression Of Timeline Of Est Start Date To Est Primary Completion Date
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Design And Rationale For A Phase Iii Randomized Placebo Controlled Trial Of Durvalumab With Or Without Tremelimumab After Concurrent Chemoradiotherapy For Patients With Limited Stage Small Cell Lung Cancer The Adriatic Study Clinical Lung
Design And Rationale For A Phase Iii Randomized Placebo Controlled Trial Of Durvalumab With Or Without Tremelimumab After Concurrent Chemoradiotherapy For Patients With Limited Stage Small Cell Lung Cancer The Adriatic Study Clinical Lung
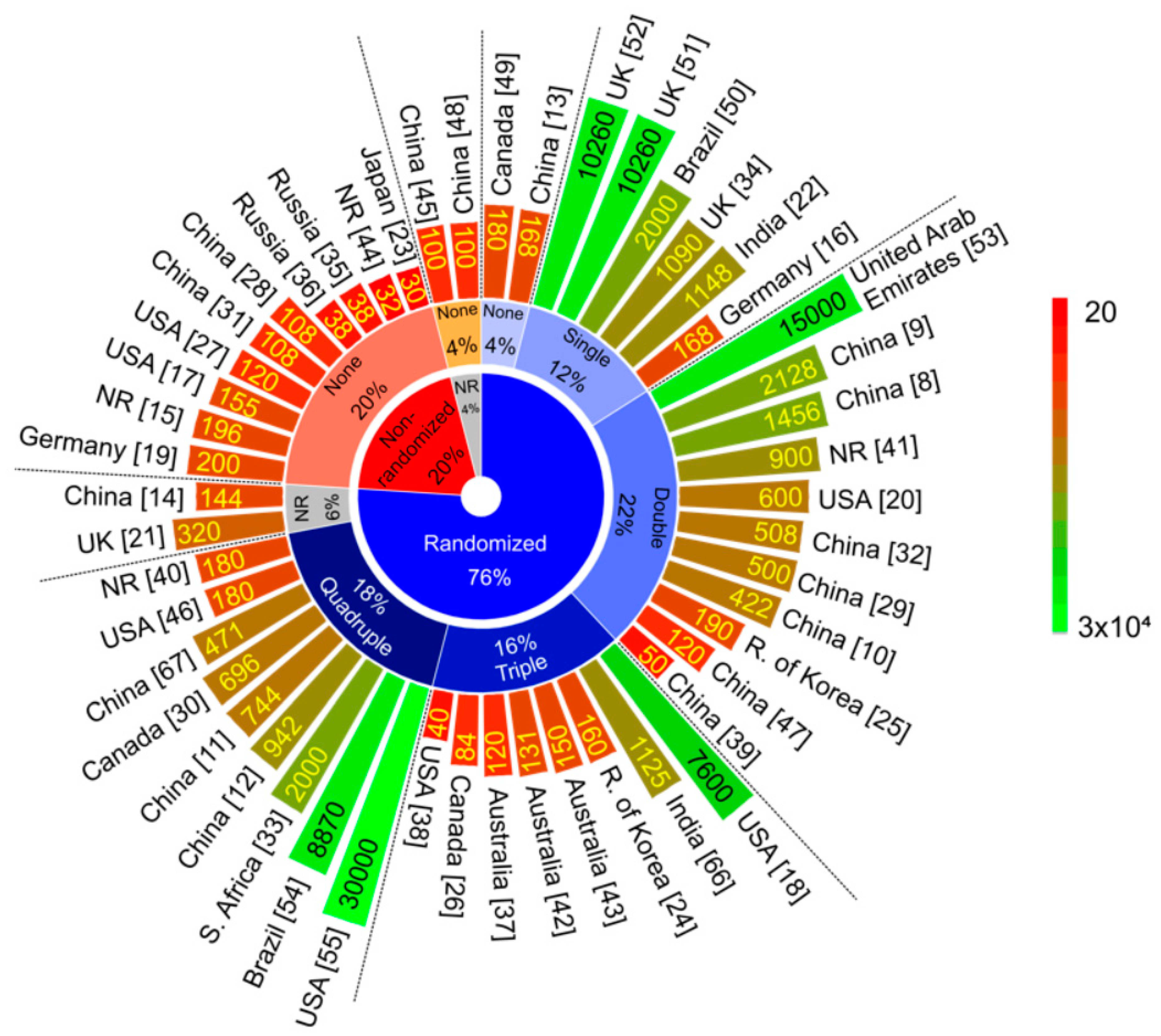 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Esmo 2019 Profound Phase 3 Study Of Olaparib Vs Enzalutamide Or Abiraterone For Metastatic Castration Resistant Prostate Cancer With Homologous Recombination Repair Gene Alterations
Esmo 2019 Profound Phase 3 Study Of Olaparib Vs Enzalutamide Or Abiraterone For Metastatic Castration Resistant Prostate Cancer With Homologous Recombination Repair Gene Alterations
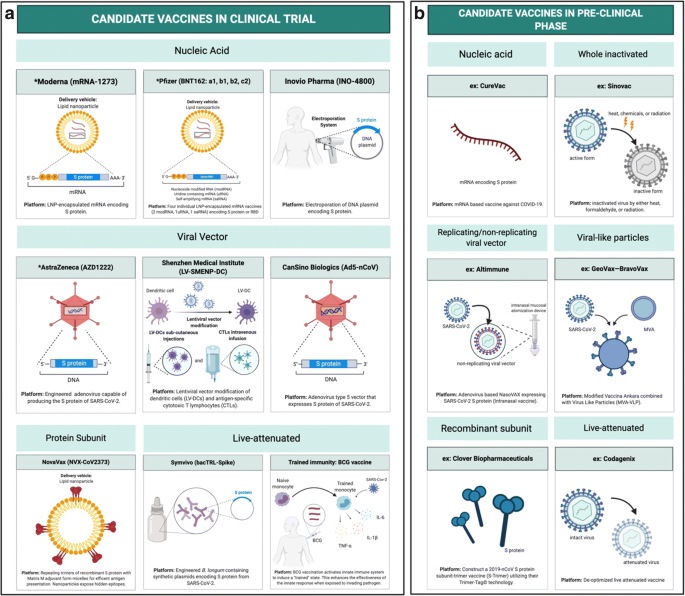 The Impact Of Immuno Aging On Sars Cov 2 Vaccine Development Springerlink
The Impact Of Immuno Aging On Sars Cov 2 Vaccine Development Springerlink
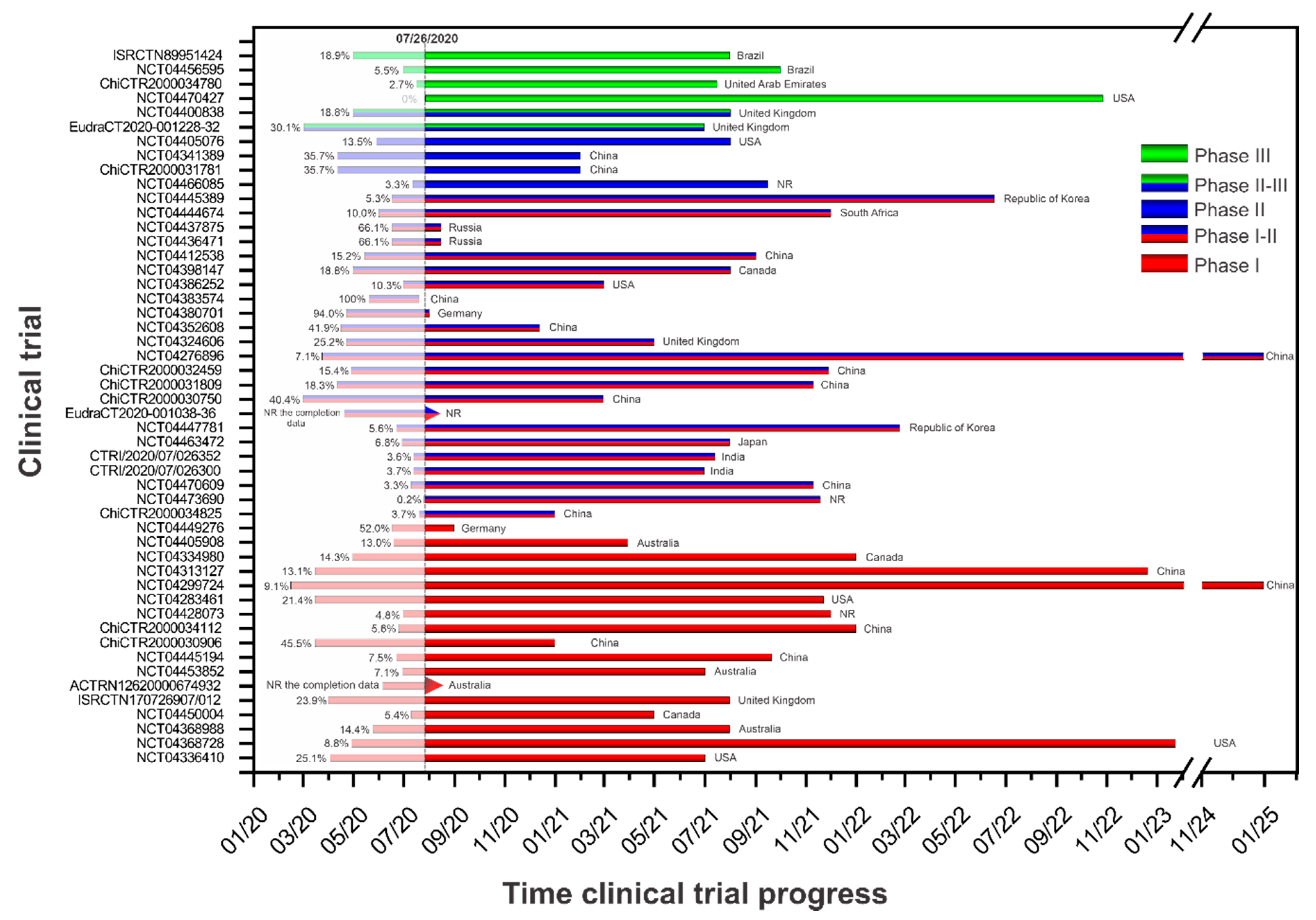 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Astrazeneca S Covid 19 Vaccine Enters Phase 3 Clinical Trial
Astrazeneca S Covid 19 Vaccine Enters Phase 3 Clinical Trial
 Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
 Oxford Astrazeneca Covid Vaccine Prompts Immune Response In All Adults
Oxford Astrazeneca Covid Vaccine Prompts Immune Response In All Adults
 Olaparib Tablets As Maintenance Therapy In Patients With Platinum Sensitive Relapsed Ovarian Cancer And A Brca1 2 Mutation Solo2 Engot Ov21 A Double Blind Randomised Placebo Controlled Phase 3 Trial The Lancet Oncology
Olaparib Tablets As Maintenance Therapy In Patients With Platinum Sensitive Relapsed Ovarian Cancer And A Brca1 2 Mutation Solo2 Engot Ov21 A Double Blind Randomised Placebo Controlled Phase 3 Trial The Lancet Oncology
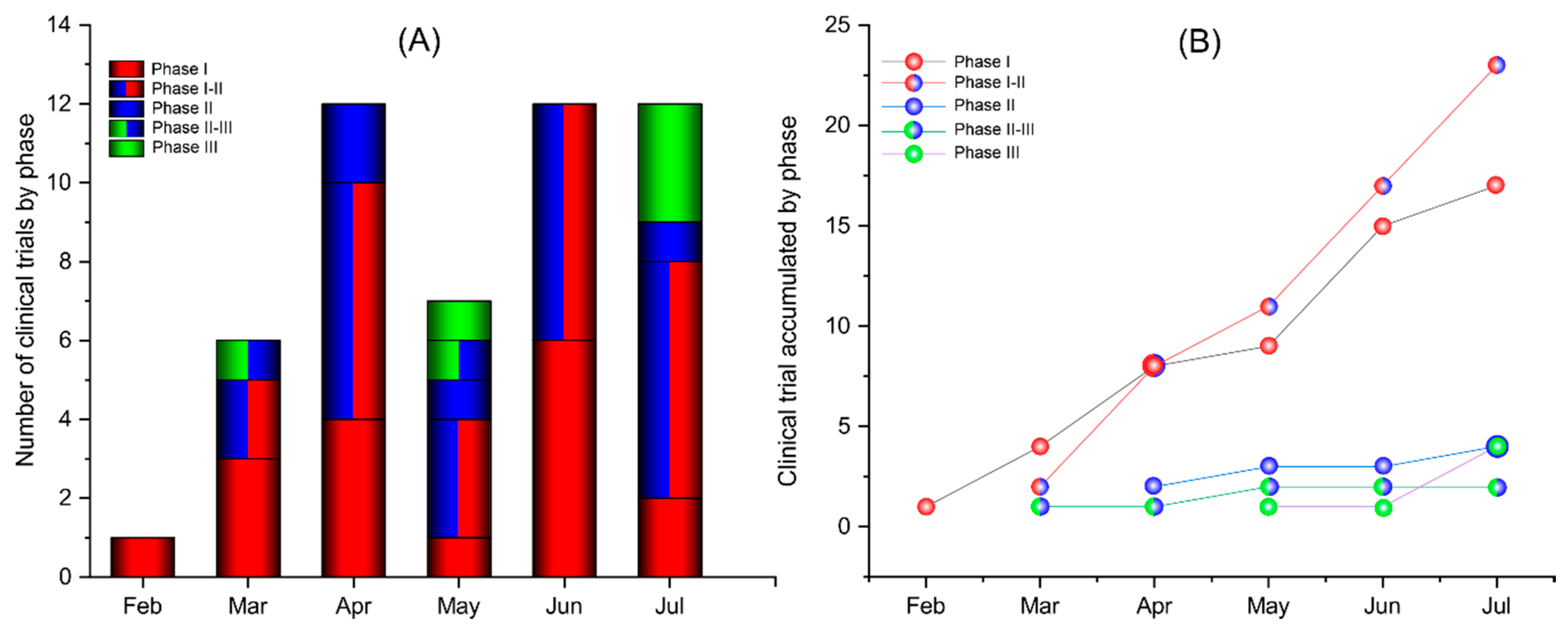 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html

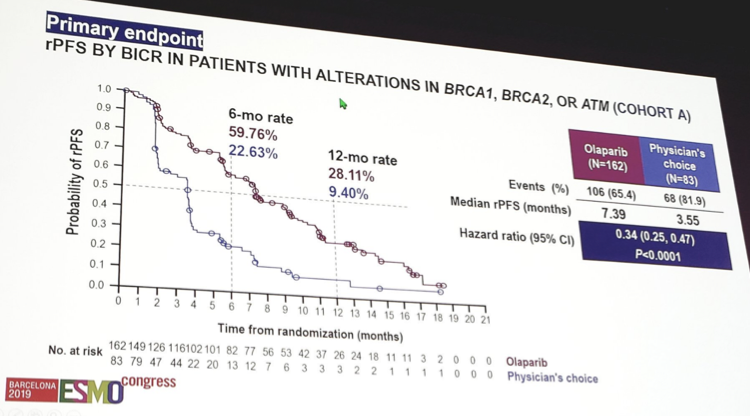 Esmo 2019 Profound Phase 3 Study Of Olaparib Vs Enzalutamide Or Abiraterone For Metastatic Castration Resistant Prostate Cancer With Homologous Recombination Repair Gene Alterations
Esmo 2019 Profound Phase 3 Study Of Olaparib Vs Enzalutamide Or Abiraterone For Metastatic Castration Resistant Prostate Cancer With Homologous Recombination Repair Gene Alterations
 A Phase Iii Study Of Triple Therapy With Budesonide Glycopyrrolate Formoterol Fumarate Metered Dose Inhaler 320 18 9 6 Mg And 160 18 9 6 Mg Using Co Suspension Delivery Technology In Moderate To Very Severe Copd The Ethos Study Protocol Respiratory
A Phase Iii Study Of Triple Therapy With Budesonide Glycopyrrolate Formoterol Fumarate Metered Dose Inhaler 320 18 9 6 Mg And 160 18 9 6 Mg Using Co Suspension Delivery Technology In Moderate To Very Severe Copd The Ethos Study Protocol Respiratory
 Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
Post a Comment for "Astrazeneca Phase 3 Study"