Astrazeneca Covid 19 Vaccine Phase 3 Results
New vaccine efficacy results are reported now in The Lancet. PfizerBioNTechs phase three trial began in late July 2020 and the results.
 Infocus Unanswered Questions On The Covid 19 Vaccine Efg Asset Management
Infocus Unanswered Questions On The Covid 19 Vaccine Efg Asset Management
Actual Study Start Date.
Astrazeneca covid 19 vaccine phase 3 results. Similar Range To Those Seen In Convalescent COVID-19 Patients In Phase III Study COV001. Added value of this study. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19.
The Oxford-AstraZeneca COVID19 vaccine is used to provide protection against infection by the SARS-CoV-2 virus in order to prevent COVID-19 in adults aged 18 years and older. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. 14092020 However some stakeholders including Pfizer which has a Phase 3 trial of its own underway say that results could be out as soon as October.
Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. 20072020 These results together with the induction of both humoral and cellular immune responses support large-scale evaluation of this candidate vaccine in an ongoing phase 3 program the researchers. And UK have published results from the final phase three trials.
We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa. Actual Primary Completion Date. Researchers at the University of Oxford have begun enrolling subjects in a phase 23 clinical trial of AstraZeneca-partnered COVID.
Cause actual outcomes and results to be materially different from those predicted. AstraZenecas COVID-19 vaccine enters phase 23 clinical trial. 20042021 The trial demonstrated that the vaccine can protect people from Covid-19 but it left many questions unresolved about the results.
AstraZeneca COVID-19 Vaccine AZD1222 ACIP COVID-19 Emergency Meeting January 27. 19012021 For these reasons even when COVID-19 vaccines have achieved licensure via current phase 3 trials there will be substantial uncertainties about how useful the vaccines will. 04022021 AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing Covid-19 more than 22 days after the first dose.
Robust Humoral Response In Older Adults Receiving AZD1222. In response to criticisms that these pharmaceutical companies are rushing to get a vaccine developed at the expense of the publics health and safety nine CEOs have signed a pledge to uphold the integrity of the scientific process as they work towards making a vaccine. An analysis published on 19 February 2021 showed an efficacy of 760 at preventing symptomatic COVID-19 beginning at 22 days following the first dose increasing to 813 when.
Estimated Study Completion Date. Reuters - An AstraZeneca AZNL executive said he felt encouraged by incredibly promising. 14042021 All four approved vaccines in the US.
Nevertheless the vaccines low cost and ease of. 20112020 AstraZeneca-Oxford COVID-19 Vaccine Shows Positive Phase II Results AZD1222 shows strong immune response fewer adverse effects in older adults November 20 2020. 22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and.
COVID-19 vaccine trial data unveiled by Pfizer and its. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19.

 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
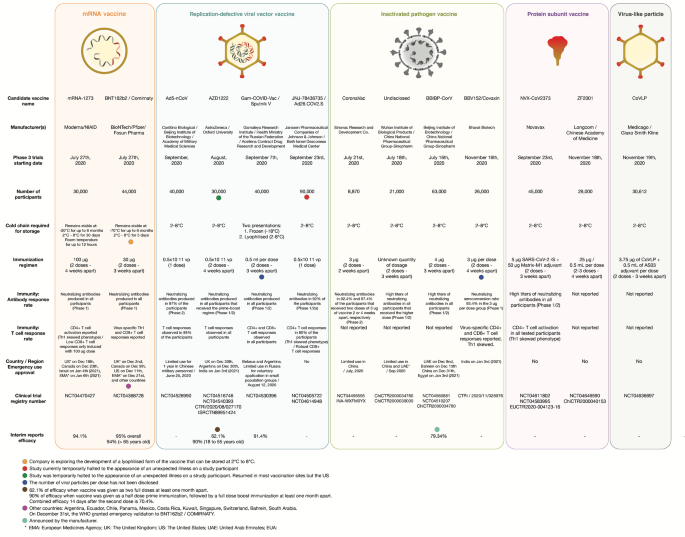 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
 Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
 Niaid Review Board Challenges New Astrazeneca Covid 19 Vaccine Data
Niaid Review Board Challenges New Astrazeneca Covid 19 Vaccine Data
 Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
 Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
 Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
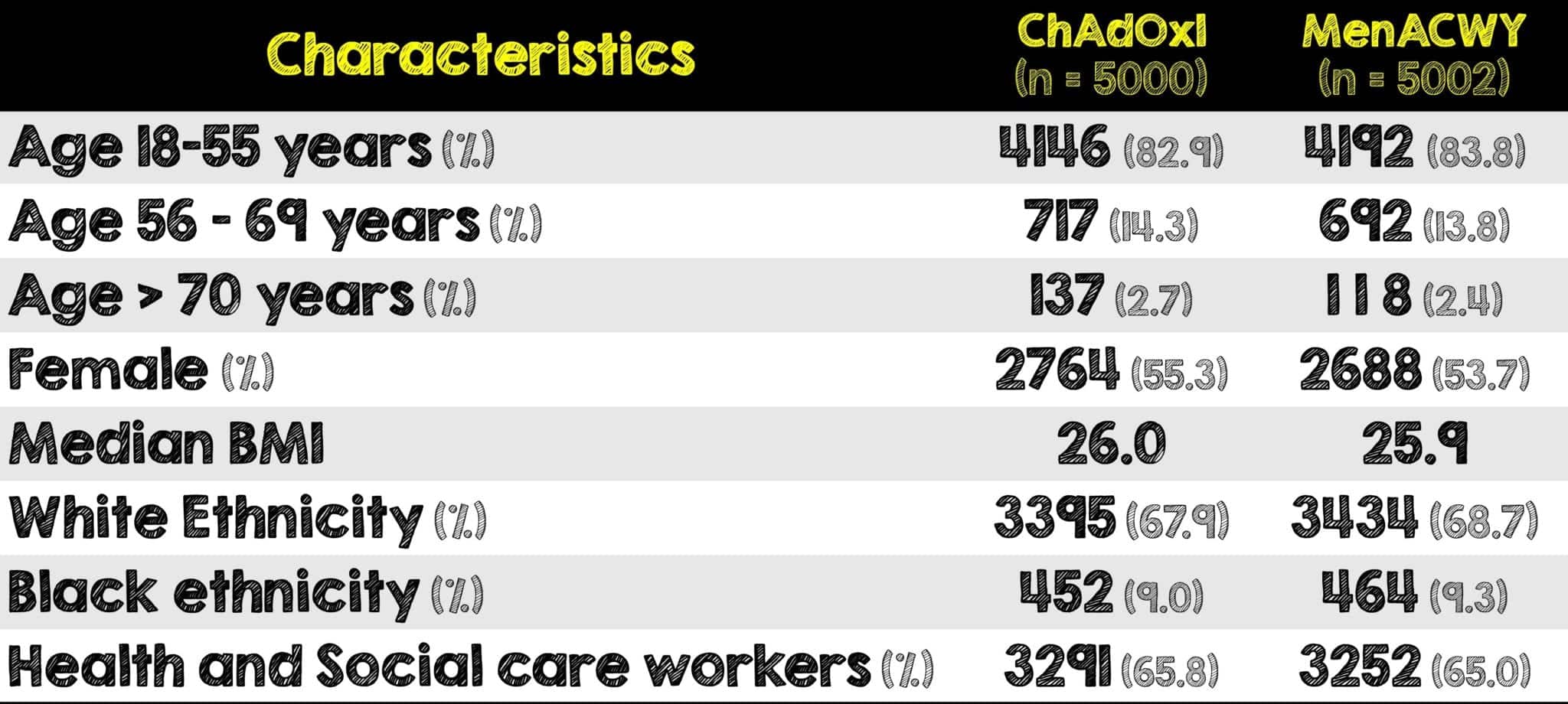 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Post a Comment for "Astrazeneca Covid 19 Vaccine Phase 3 Results"