Astrazeneca Vaccine Phase 3 Trial Results
Estimated Study Completion Date. Vaccine immunogenicity efficacy and safety were demonstrated in four Phase I-III non-IND trials in UK Brazil.
 Astrazeneca Oxford University Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Astrazeneca Oxford University Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa.

Astrazeneca vaccine phase 3 trial results. AstraZeneca committed to a partnership with Oxford University to ensure broad and equitable vaccine access globally not for profit during the pandemic. 20072020 According to AstraZeneca late-stage trials of AZD1222 include a Phase III trial in Brazil 2000 participants ISRCTN89951424 a Phase IIbIII trial in. The interim analysis for efficacy was based on 11636 participants accruing 131.
This Roundup accompanied an SMC Briefing. 30112020 Modernas phase 3 analysis involved 30000 people in the US half given the vaccine and half a placebo and 196 of them became ill. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older.
New vaccine efficacy results are reported now in The Lancet. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US.
19072020 Brazil and India follow with a staggering 201 million and 1 million confirmed cases respectively. Following impressive phase 3 clinical trial results from Pfizer Inc. AstraZeneca and the University of Oxford released phase 3 clinical.
The company now says. 03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose. 05112020 Pharmaceutical company AstraZeneca said Wednesday that phase three trial results for its COVID-19 vaccine could be ready later this year.
The University of Oxford. Oxford COVID-19 vaccine to begin phase IIIII human trials. The results of the phase 3 clinical trials of the Oxford AstraZeneca COVID-19 vaccine candidate have been published in The Lancet.
Purportedly out-of-date data in the original phase 3 preliminary study AstraZeneca conducted a. 22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and. Prof Sheila Bird Formerly Programme Leader MRC Biostatistics Unit University of Cambridge said.
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. Actual Study Start Date. 26032021 Home Coronavirus News AstraZeneca COVID-19 vaccine trial data results in.
Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation. First peer-reviewed results of phase 3 human trials of Oxford coronavirus vaccine demonstrate efficacy University of Oxford. And Moderna Inc AstraZeneca was in the spotlight today.
Actual Primary Completion Date. 04022021 AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing Covid-19 more than 22 days after the first dose. Oxford and AstraZeneca researchers present a pooled analysis in The Lancet of Phase 3 trials of a coronavirus vaccine resulting in an average efficacy of 704.
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Oxford Covid 19 Vaccine Shows 70 Efficacy In Phase 2 3 Trials Hindustan Times
Oxford Covid 19 Vaccine Shows 70 Efficacy In Phase 2 3 Trials Hindustan Times
 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
 Oxford Astrazeneca Covid 19 Vaccine 5 Sites Selected For Phase 3 Clinical Trial In India
Oxford Astrazeneca Covid 19 Vaccine 5 Sites Selected For Phase 3 Clinical Trial In India
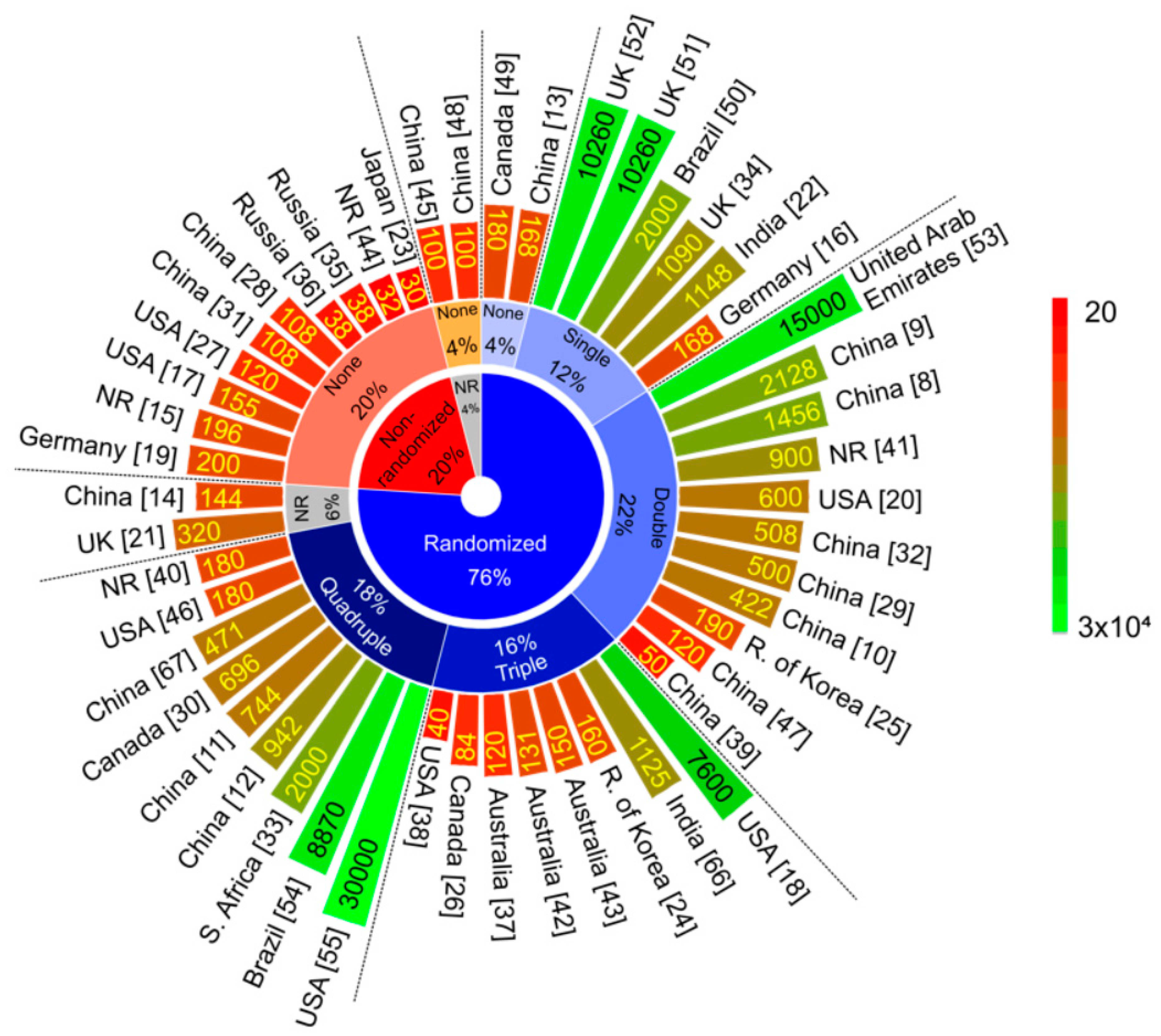 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
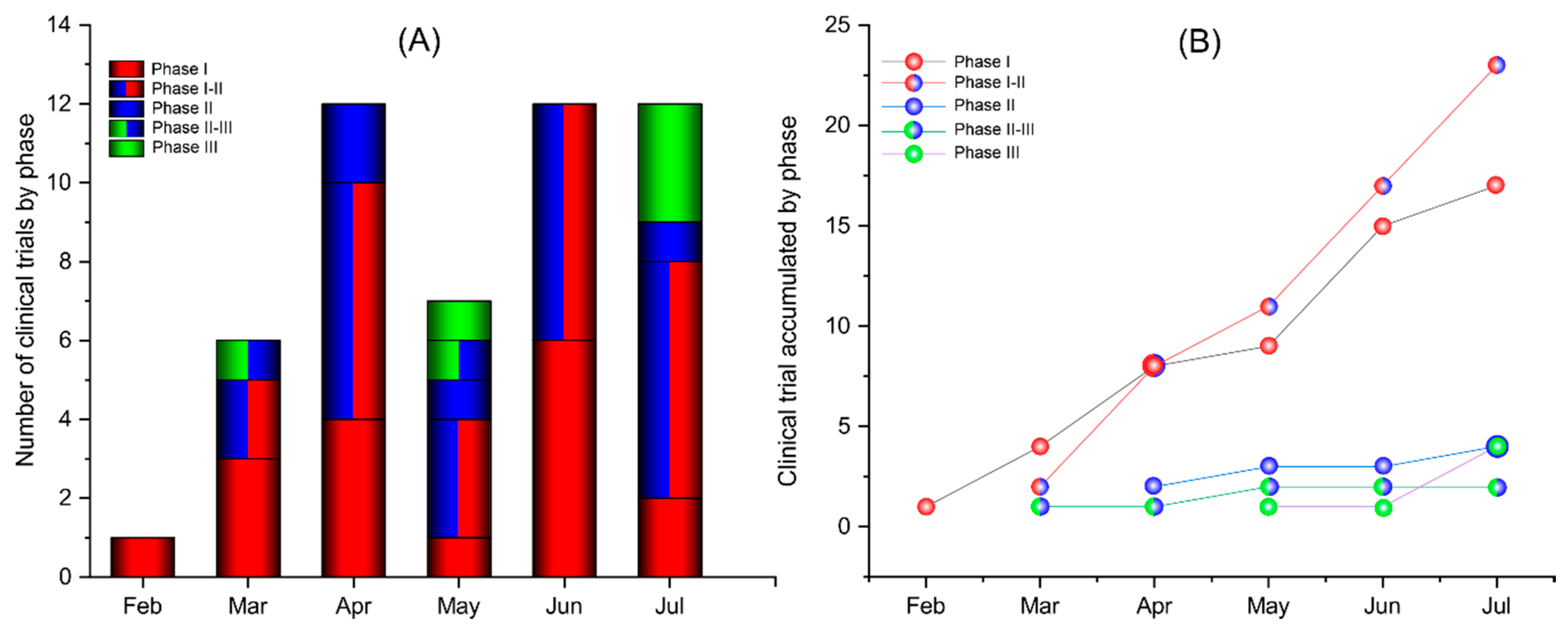 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html

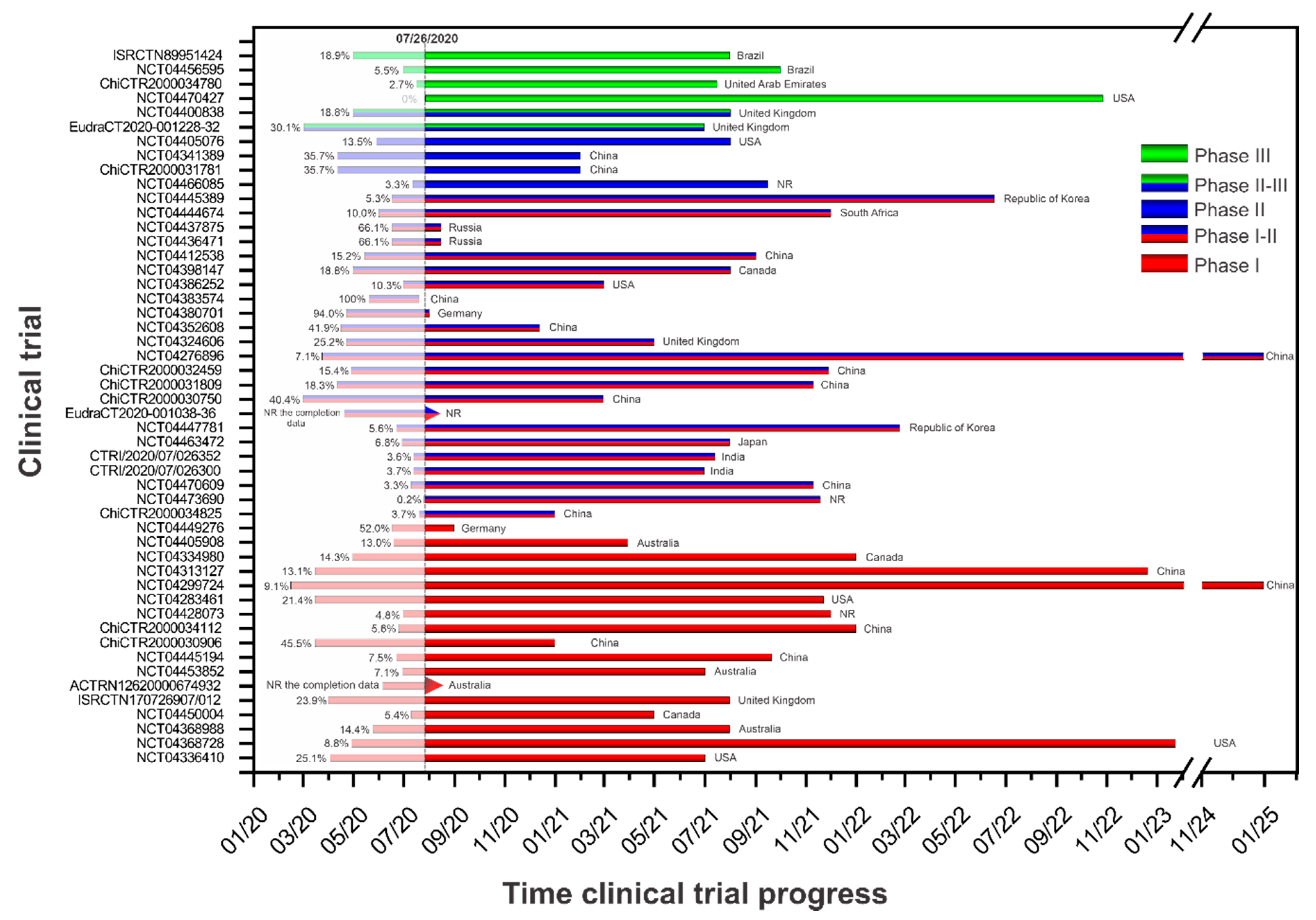 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Safety And Efficacy Of An Rad26 And Rad5 Vector Based Heterologous Prime Boost Covid 19 Vaccine An Interim Analysis Of A Randomised Controlled Phase 3 Trial In Russia The Lancet
Safety And Efficacy Of An Rad26 And Rad5 Vector Based Heterologous Prime Boost Covid 19 Vaccine An Interim Analysis Of A Randomised Controlled Phase 3 Trial In Russia The Lancet
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
 Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
Post a Comment for "Astrazeneca Vaccine Phase 3 Trial Results"