Astrazeneca Covid 19 Vaccine Phase 3
The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19. We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa.
 Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
14092020 AstraZeneca one of the pharmaceutical companies developing a COVID-19 vaccine announced over the weekend that clinical trials in the UK.
Astrazeneca covid 19 vaccine phase 3. The next stage of. AstraZeneca has begun a US. In a virtual presser DOST-Philippine Council for Health Research and Development executive.
AstraZeneca today said its AZD1222 COVID-19 vaccine candidate is safe and effective based on data culled from a phase 3 clinical trial of 32449 US. The British-Swedish pharma company AstraZeneca has temporarily halted phase 3 clinical trials of the AZD1222 COVID-19 vaccine after a trial participant reportedly developed severe. Actual Study Start Date.
The company got the green light from the Medicines Health Regulatory Authority that continuing to test the vaccine. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. The vaccine was initially developed by the University of Oxford but AstraZeneca will manufacture and distribute the vaccine worldwide.
20072020 Researchers from AstraZeneca the University of Oxford and its spinout company Vaccitech have published preliminary Phase III data showing their closely-watched COVID-19 vaccine candidate. 19012021 For these reasons even when COVID-19 vaccines have achieved licensure via current phase 3 trials there will be substantial uncertainties about how useful the vaccines will. Phase-3 trial of Oxford-AstraZeneca Covid-19 vaccine to begin in few days.
The drug maker said its two-dose candidate is 79 effective in preventing symptomatic COVID-19 with a 100 efficacy against severe or critical disease and hospitalization. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. The AstraZeneca COVID-19 vaccine is the third vaccine over the past few.
Actual Primary Completion Date. 22032021 Big relief for AstraZeneca. Volunteers can be given the next dose only 28 days after completing the.
Phase 3 Clinical Testing in the US. The Biomedical Advanced Research and Development Authority-funded study will enroll 30000 adults at. New vaccine efficacy results are reported now in The Lancet.
The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. 30112020 Screenshot from the Laging Handa public briefing on Nov. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature.
Estimated Study Completion Date. 23112020 AstraZeneca has announced that its COVID-19 vaccine has exhibited over 70 average effectiveness in phase 3 clinical trials in Brazil and the United Kingdom. Researchers at the University of Oxford have begun enrolling subjects in a phase 23 clinical trial of AstraZeneca-partnered COVID-19 vaccine AZD1222.
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704. MANILA Pharmaceutical firm AstraZeneca would like to conduct a Phase 3 clinical trial in the Philippines an official of the Department of Science and Technology DOST said on Monday.
Phase 3 clinical trial of its COVID-19 vaccine candidate AZD1222. COVID-19 vaccine shows 79 efficacy in US Phase-3 trials The interim safety and efficacy analysis was based on 32449 participants accruing 141 symptomatic cases of COVID-19.
 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021


 Astrazeneca S Covid 19 Vaccine Enters Phase 3 Clinical Trial
Astrazeneca S Covid 19 Vaccine Enters Phase 3 Clinical Trial
 Covid 19 Vaccine Trial Will Continue After Volunteer Death Science Business
Covid 19 Vaccine Trial Will Continue After Volunteer Death Science Business
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
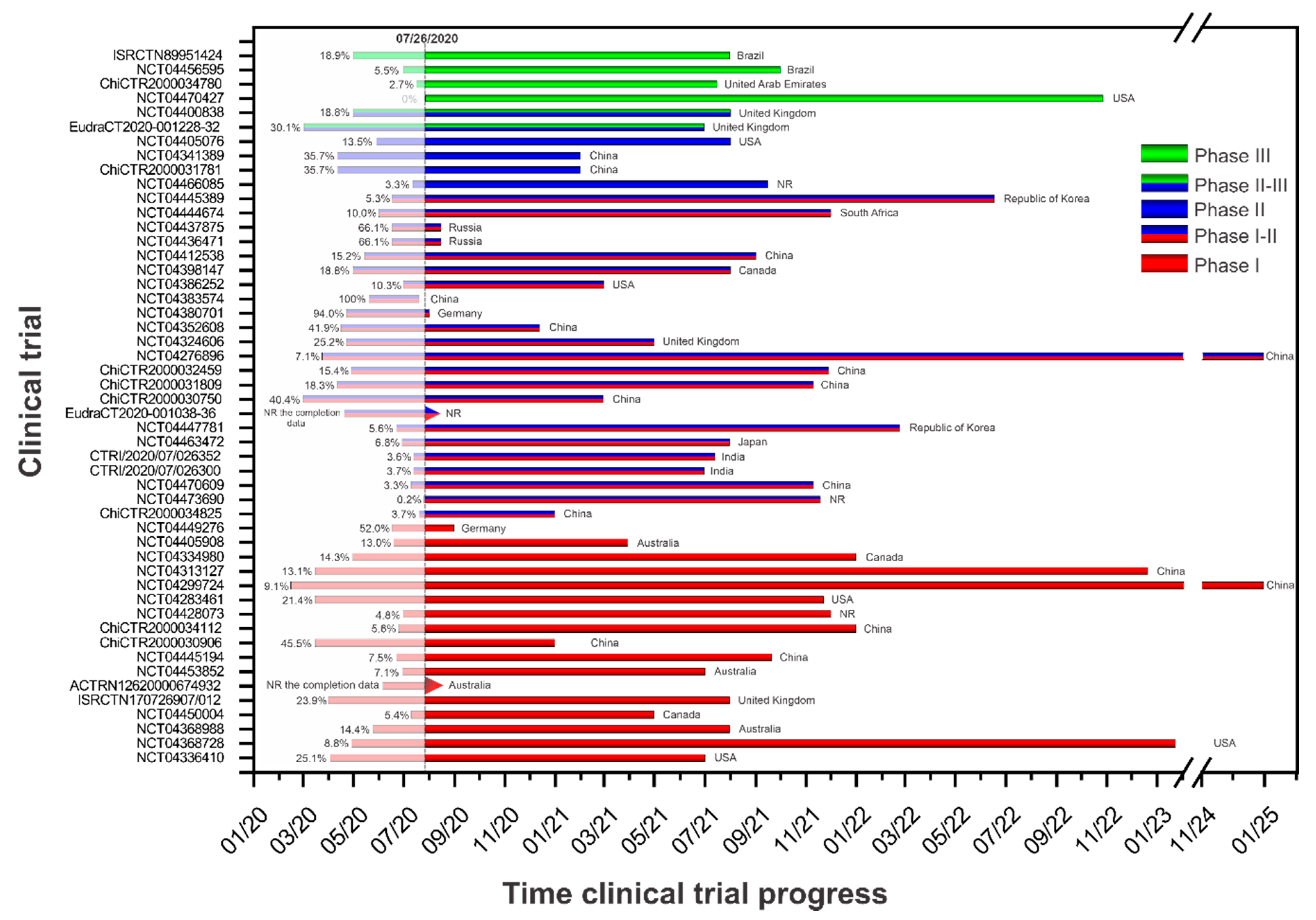 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post
Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post
 Covid 19 Vaccine These Countries Have Started Rolling Out Coronavirus Vaccine Check Full List Here
Covid 19 Vaccine These Countries Have Started Rolling Out Coronavirus Vaccine Check Full List Here
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India
Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India
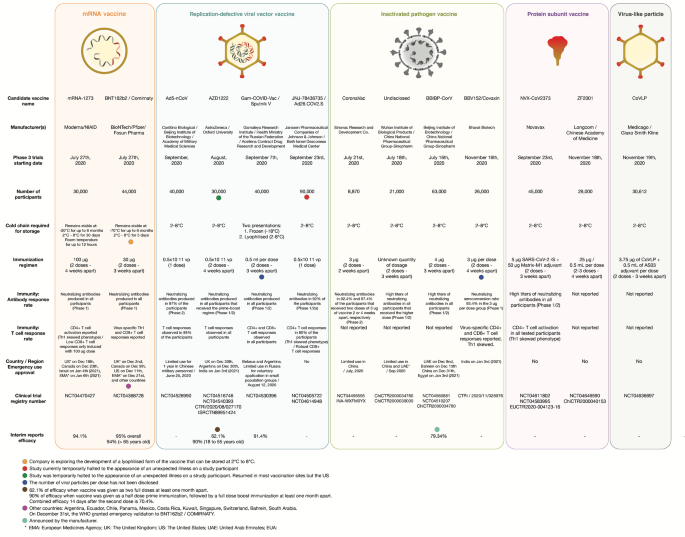 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
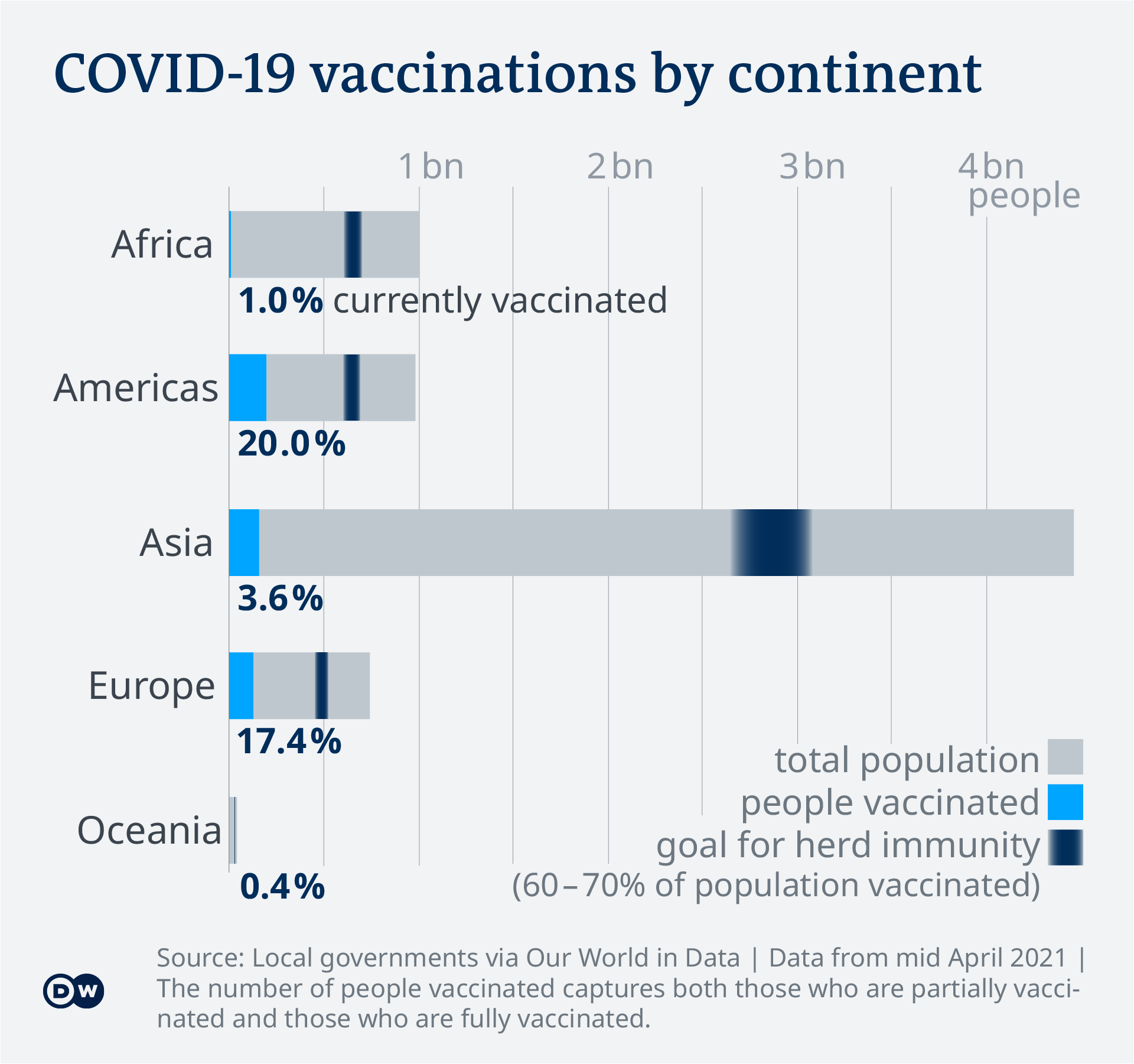 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
 Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
 Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Post a Comment for "Astrazeneca Covid 19 Vaccine Phase 3"