Astrazeneca Phase 3 Trial Date
03022021 COVID-19 Vaccine AstraZeneca confirms 100 protection against severe disease hospitalisation and death in the primary analysis of Phase III trials. 1 min read AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety.
Imfinzi Plus Tremelimumab Demonstrated Promising Clinical Activity And Tolerability In Patients With Advanced Liver Cancer
A release issued on Monday reported a symptomatic efficacy rate of 79.
Astrazeneca phase 3 trial date. The company now says its vaccine is 76 effective in protecting against symptomatic cases of virus. The interim analysis published today in. In the case of the prominent Moderna mRNA-1273 trial the start date of the Phase 3 study was July 27 2020.
But what about the promising BNT162 trial. 25032021 March 24 2021 947 PM. Now the estimated primary completion date is actually October 27 2021 a little over a year from the start date.
AstraZeneca currently has numerous clinical trials in progress around the world for its. Actual Study Start Date. 25032021 AstraZeneca issued updated Phase 3 trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report.
This analysis includes data from four ongoing blinded randomised controlled trials done across the UK Brazil and South Africa. Because we operate in an ever-changing environment. Trial found its vaccine.
Published online Nov 19. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults COV002. April 26 2021 at 218 am.
Trial participants to date are aged 18 years or over who are healthy or have medically stable chronic diseases and are at increased risk for being exposed to the SARS-CoV-2 virus. The new study published in the Lancet is the first peer-reviewed publication of phase 3 data from studies of a vaccine against the coronavirus. Estimated Study Completion Date.
25032021 AstraZeneca on Thursday published an updated analysis confirming that a Phase 3 trial involving its COVID-19 vaccine was safe and effective and. Participants are randomised to receive two intramuscular doses of a full dose 5x10 10 viral particles of AZD1222 or comparator meningococcal vaccine MenACWY as first dose and a saline placebo as. PUBLISHED 3 February 2021.
The company reported on Monday that a US. A single-blind randomised controlled phase 23 trial. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
And Brazil and in a Phase 12 trial in South Africa. Published 8 Dec 2020. Participants aged 18 years and older were randomly assigned 11 to ChAdOx1 nCoV-19 vaccine or control meningococcal group A C W and Y.
19012021 The FDA guidance for Emergency Use Authorization suggests a median duration of follow-up of phase 3 vaccine trial volunteers of 2 months 43. This web site provides clinical trial data results and other information from or regarding AstraZeneca-sponsored clinical trials. With a 76 efficacy rate against symptomatic COVIDFlashback.
Most events are expected to fall within that window. Investigators recently reported promising results in The Lancet. And the estimated final end date is the same.
This site is part of our commitment to provide patients and healthcare professionals with meaningful information about AstraZeneca medicines and drugs in development. 13042021 AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety. 26042021 AstraZenecas Phase 3 Nirsevimab trial successful Published.
But before I describe this new data some quick background info is in order. Actual Primary Completion Date. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19.
Based on the RML data a Phase 1 trial of the candidate vaccine began on April 23 in healthy volunteers in the UK. Double-blind placebo-controlled data has finally been published for the Oxford-AstraZeneca COVID-19 vaccine - and it shows the vaccine does not prevent infection with the South African variant of COVID-19. Currently the vaccine candidate is being evaluated in Phase 23 trials in the UK.
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 The Lead Covid 19 Vaccine Candidates Where We Are And What You Should Know Lexology
The Lead Covid 19 Vaccine Candidates Where We Are And What You Should Know Lexology
 A Phase Iii Study Of Triple Therapy With Budesonide Glycopyrrolate Formoterol Fumarate Metered Dose Inhaler 320 18 9 6 Mg And 160 18 9 6 Mg Using Co Suspension Delivery Technology In Moderate To Very Severe Copd The Ethos Study Protocol Respiratory
A Phase Iii Study Of Triple Therapy With Budesonide Glycopyrrolate Formoterol Fumarate Metered Dose Inhaler 320 18 9 6 Mg And 160 18 9 6 Mg Using Co Suspension Delivery Technology In Moderate To Very Severe Copd The Ethos Study Protocol Respiratory
 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
 Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
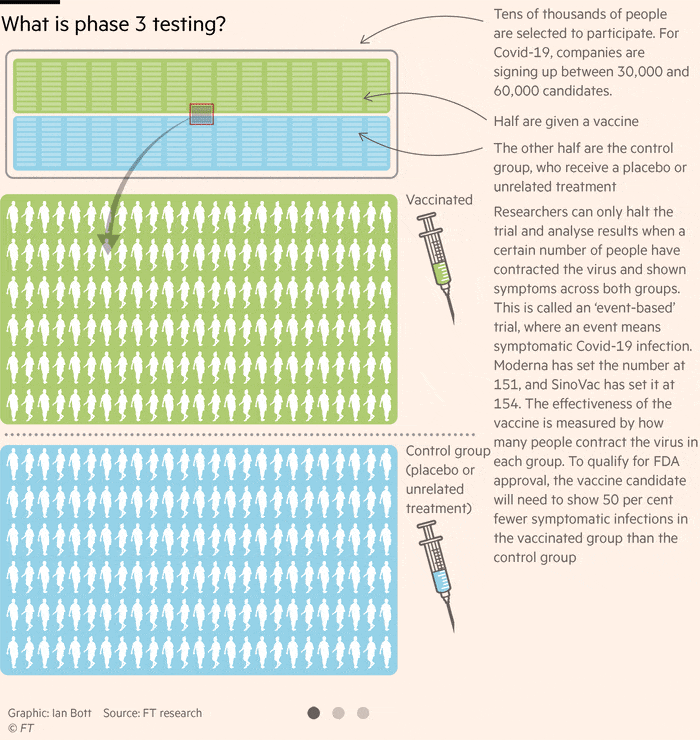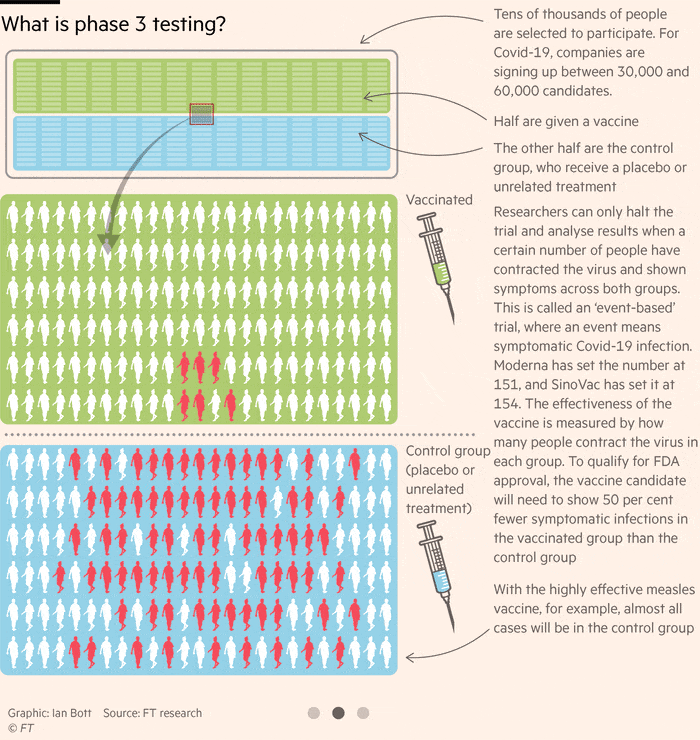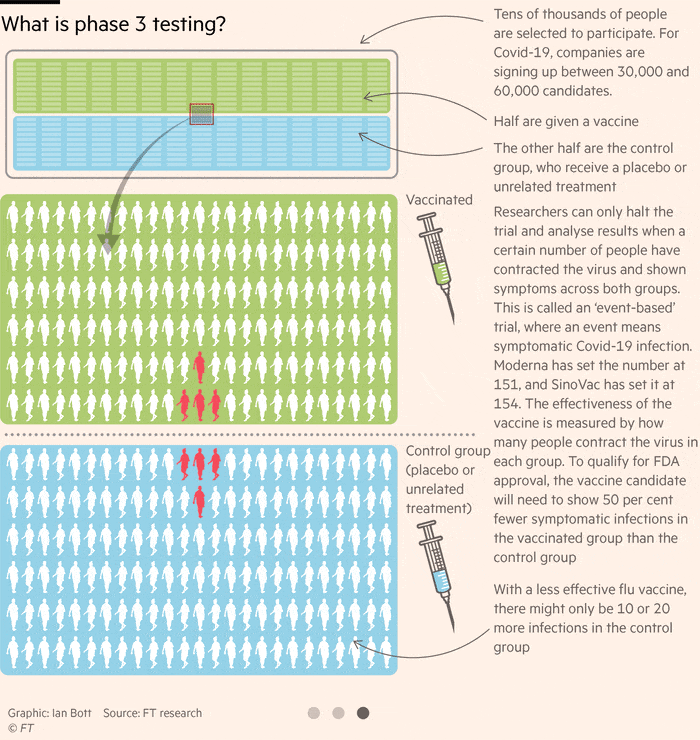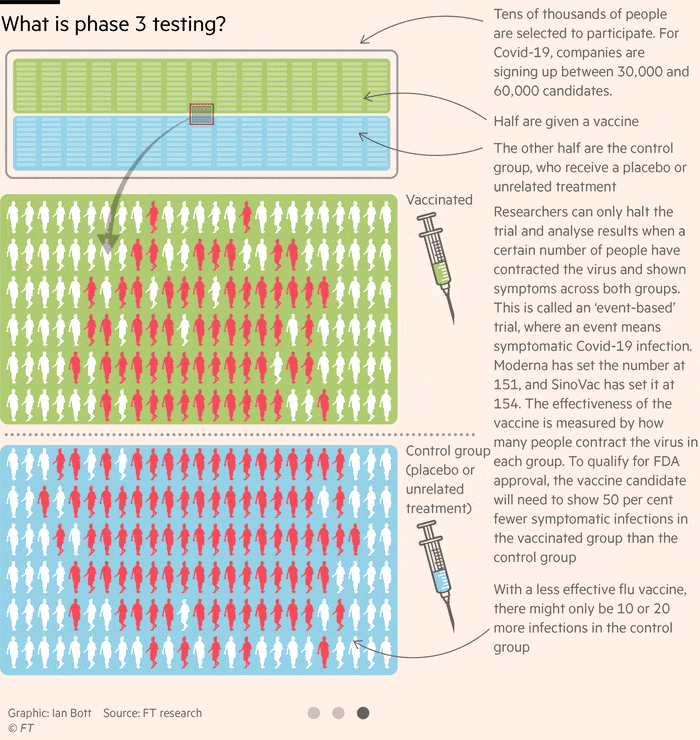 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Astrazeneca Vaccine Document Shows Limit Of No Profit Pledge Financial Times
Astrazeneca Vaccine Document Shows Limit Of No Profit Pledge Financial Times
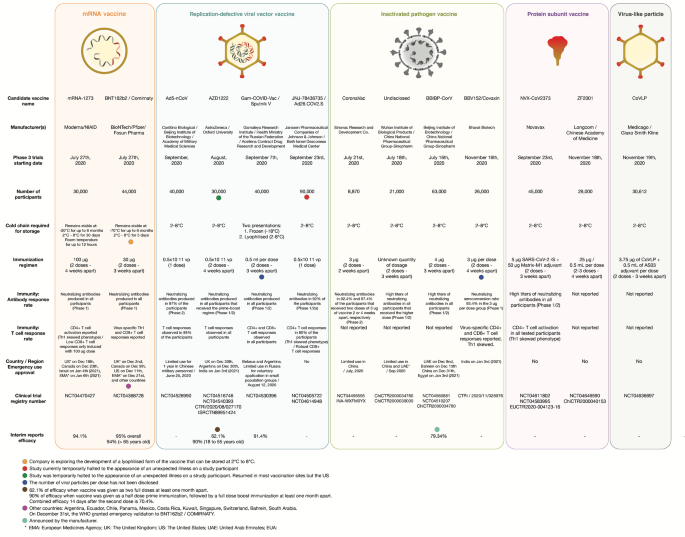 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines

 Covid 19 Vaccines November Update Progress Of Clinical Trials Post
Covid 19 Vaccines November Update Progress Of Clinical Trials Post
 Oxford Astrazeneca Covid 19 Vaccine 5 Sites Selected For Phase 3 Clinical Trial In India
Oxford Astrazeneca Covid 19 Vaccine 5 Sites Selected For Phase 3 Clinical Trial In India
 Olaparib Tablets As Maintenance Therapy In Patients With Platinum Sensitive Relapsed Ovarian Cancer And A Brca1 2 Mutation Solo2 Engot Ov21 A Double Blind Randomised Placebo Controlled Phase 3 Trial The Lancet Oncology
Olaparib Tablets As Maintenance Therapy In Patients With Platinum Sensitive Relapsed Ovarian Cancer And A Brca1 2 Mutation Solo2 Engot Ov21 A Double Blind Randomised Placebo Controlled Phase 3 Trial The Lancet Oncology
Post a Comment for "Astrazeneca Phase 3 Trial Date"