Astrazeneca Vaccine 30 Cases
Alberto PezzaliThe Associated Press British regulators on Thursday said they have identified 30 cases of. 03042021 In total MHRA said had identified 30 cases of rare blood clot events out of 181 million AstraZeneca doses administered up to and including March.
 Explainer How Worried Should We Be About Reports Of Blood Clots And Astrazeneca S Vaccine Reuters
Explainer How Worried Should We Be About Reports Of Blood Clots And Astrazeneca S Vaccine Reuters
03042021 As many as 30 cases of rare blood clot events have been identified after the use of the AstraZeneca COVID-19 vaccine British regulators on.
Astrazeneca vaccine 30 cases. 02042021 British regulators on Thursday said they have identified 30 cases of rare blood clot events after the use of the AstraZeneca COVID-19 vaccine 25. The Medicines and Healthcare products Regulatory Agency. 02042021 Among those who received that AstraZeneca coronavirus vaccine doses only around 30 cases of blood clots were reported till March 24 including 22 cases of cerebral vein thrombosis and eight other.
The United Kingdom has reported 30 cases of blood clots after the use of the AstraZeneca vaccine against coronavirus. 02042021 London UK April 2 ANI. 08042021 Under 30s who have already had their first dose of the OxfordAstraZeneca vaccine are being urged to come forward for their second jab despite concerns over a.
03042021 In total MHRA said it had identified 30 cases of rare blood clot events out of 181 million AstraZeneca doses administered up to and including March 24. 02042021 A vial of the AstraZeneca-Oxford COVID-19 vaccine is seen in Luton England on March 21. Thirty cases of rare blood clotting have been recorded in Britain among more than 18 million people who have had the AstraZeneca COVID-19 vaccine the national medicines regulator said.
03042021 The UKs medicines watchdog says it has found 30 cases of rare blood clots in people who have had the Oxford-AstraZeneca Covid vaccine. 02042021 Reuters - British regulators on Thursday said they have identified 30 cases of rare blood clot events after the use of the AstraZeneca COVID. EMA will further communicate as the assessment progresses.
Has recorded 30 cases of rare blood clotting events after the use of the AstraZeneca Covid-19 vaccine the countrys drug regulator announced on Thursday evening but insisted that the. The Medicines and Healthcare products Regulatory Agency said it had received no such reports of clotting events following use of the vaccine made by BioNTech SE and Pfizer Inc. The British health officials said the benefits of the.
02042021 Britains medicines regulator has reported that 30 people who received the Oxford-AstraZeneca vaccine developed rare blood clots up from five. 06042021 British health officials said there have been 30 cases of blood clots after the use of the AstraZeneca COVID-19 vaccine according to Reuters. 01042021 British regulators on Thursday said they have identified 30 cases of rare blood clot events after the use of the AstraZeneca COVID-19 vaccine 25.
As of 10 March 2021 30 cases 1 of thromboembolic events had been reported among close to 5 million people vaccinated with COVID-19 Vaccine AstraZeneca in the European Economic Area. 02042021 British regulators said they have identified 30 cases of rare blood clots associated with the AstraZeneca vaccine but stressed the benefits of the jab in. 02042021 British regulators on Thursday said they have identified 30 cases of rare blood clot events after the use of the AstraZeneca COVID-19 vaccine 25 more than the agency previously reported.
02042021 Britain reported 30 cases of extremely rare blood clots in people who had received the AstraZeneca-Oxford vaccine the same sort of events that have prompted some European countries to restrict use. British regulators have found a total of 30 cases of blood clot events after the use of the AstraZeneca vaccine Regulators in the UK have identified 30 cases of rare blood clot events.
 Astrazeneca Jab Mhra And Ema To Give Update On Blood Clot Investigation Vaccines And Immunisation The Guardian
Astrazeneca Jab Mhra And Ema To Give Update On Blood Clot Investigation Vaccines And Immunisation The Guardian
 Astrazeneca Ema Says Blood Clots Very Rare Side Effect News Dw 07 04 2021
Astrazeneca Ema Says Blood Clots Very Rare Side Effect News Dw 07 04 2021
 South Korea To Resume Astrazeneca Vaccinations For Ages 30 60 Coronavirus Outbreak News
South Korea To Resume Astrazeneca Vaccinations For Ages 30 60 Coronavirus Outbreak News
 Britain Sees 25 New Blood Clot Cases Linked To Astrazeneca Vaccine Business Standard News
Britain Sees 25 New Blood Clot Cases Linked To Astrazeneca Vaccine Business Standard News
 Germany Restricts Use Of Astrazeneca Vaccine To Over 60s In Most Cases News Dw 30 03 2021
Germany Restricts Use Of Astrazeneca Vaccine To Over 60s In Most Cases News Dw 30 03 2021
 What You Need To Know About Astrazeneca S Covid 19 Vaccine Science In Depth Reporting On Science And Technology Dw 18 03 2021
What You Need To Know About Astrazeneca S Covid 19 Vaccine Science In Depth Reporting On Science And Technology Dw 18 03 2021
 Mhra Reviewing Astrazeneca Vaccine Use In Under 30s Pulse Today
Mhra Reviewing Astrazeneca Vaccine Use In Under 30s Pulse Today
 Coronavirus Digest Uk Finds 30 Blood Clot Cases After Astrazeneca Jab News Dw 02 04 2021
Coronavirus Digest Uk Finds 30 Blood Clot Cases After Astrazeneca Jab News Dw 02 04 2021
 Uk Regulator Reports 30 Cases Of Rare Blood Clot Events After Astrazeneca Jab South Wales Argus
Uk Regulator Reports 30 Cases Of Rare Blood Clot Events After Astrazeneca Jab South Wales Argus
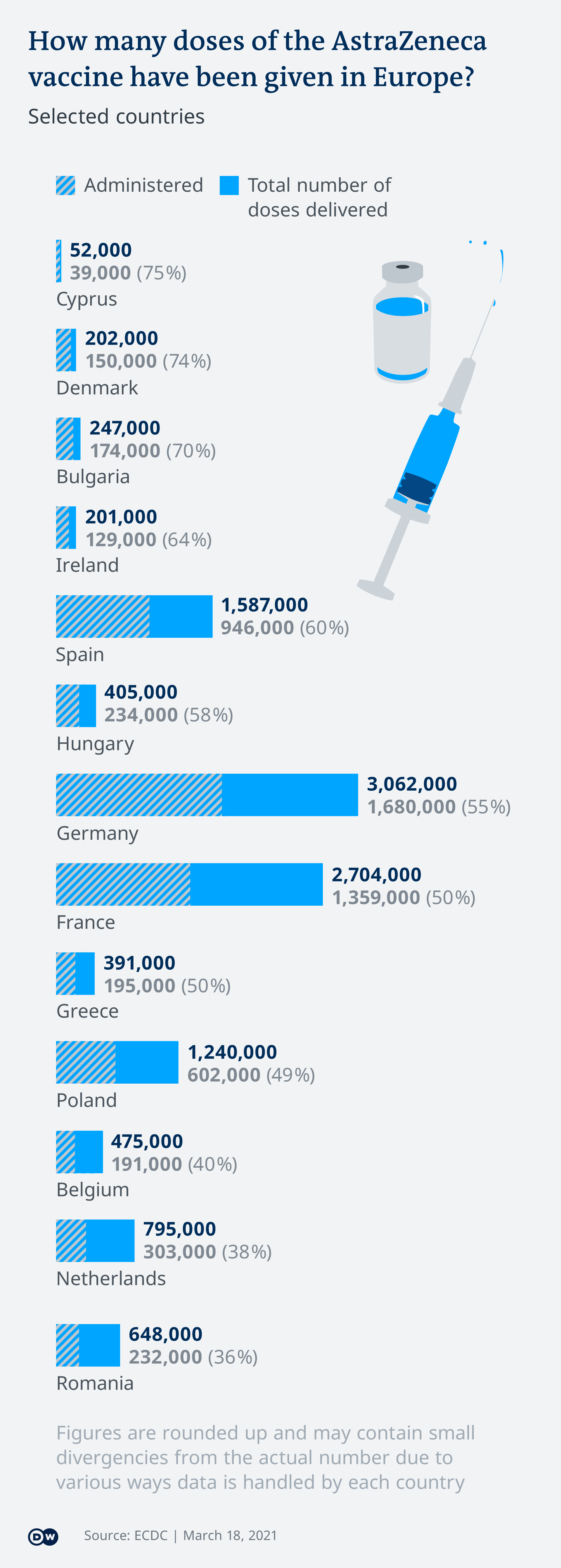 Germany Restricts Use Of Astrazeneca Vaccine To Over 60s In Most Cases News Dw 30 03 2021
Germany Restricts Use Of Astrazeneca Vaccine To Over 60s In Most Cases News Dw 30 03 2021
 Uk Reports Five Cases Of Blood Clotting In Brain Among Astrazeneca Vaccine Recipients Al Arabiya English
Uk Reports Five Cases Of Blood Clotting In Brain Among Astrazeneca Vaccine Recipients Al Arabiya English
 Oxford Astrazeneca Vaccine Two Views On Blood Clot Concerns Coronavirus The Guardian
Oxford Astrazeneca Vaccine Two Views On Blood Clot Concerns Coronavirus The Guardian
 Nigeria Pivots From Astrazeneca To Johnson Johnson Covid 19 Vaccine Health Policy Watch
Nigeria Pivots From Astrazeneca To Johnson Johnson Covid 19 Vaccine Health Policy Watch
 Eu Regulator Astrazeneca Vaccine Effective Blood Clots May Be A Rare Side Effect Coronavirus Updates Npr
Eu Regulator Astrazeneca Vaccine Effective Blood Clots May Be A Rare Side Effect Coronavirus Updates Npr
 Ema Sees No Evidence To Restrict Astrazeneca Vaccine News Dw 31 03 2021
Ema Sees No Evidence To Restrict Astrazeneca Vaccine News Dw 31 03 2021
 Q A Is The Astrazeneca Covid 19 Vaccine Safe For Older People
Q A Is The Astrazeneca Covid 19 Vaccine Safe For Older People
 U K Regulator Found 30 Cases Of Blood Clot Events After Astrazeneca Vaccine Use The Japan Times
U K Regulator Found 30 Cases Of Blood Clot Events After Astrazeneca Vaccine Use The Japan Times


Post a Comment for "Astrazeneca Vaccine 30 Cases"