Astrazeneca Vaccine Phase 3 Results Date
Based on the RML data a Phase 1 trial of the candidate vaccine began on April 23 in healthy volunteers in the UK. 11 February 2021 904 am.
 Latest U S Will Share Its Astrazeneca Vaccine Supply With The World
Latest U S Will Share Its Astrazeneca Vaccine Supply With The World
31 Jul 2020 Last Updated July 31st 2020 0715 AstraZeneca has reported that it has obtained good data so far on its Covid-19 vaccine candidate which was licensed from the University of Oxford and is currently in large-scale clinical studies.
Astrazeneca vaccine phase 3 results date. AstraZeneca COVID Vaccine 79 Efficacious in Phase 3 Interim Results - Medscape - Mar 22 2021. Actual Study Start Date. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature.
Tuesdays results the first Phase 3 clinical data for a. We will continue to work with our partners and governments around the world to prepare for global distribution in 2020 and beyond The Phase 3 clinical trial of BNT162b2 began on July 27 and has enrolled 43661 participants to date 41135 of whom have received a second dose of the vaccine candidate as of November 13 2020. 3-min read AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US.
25032021 March 24 2021 947 PM. Actual Primary Completion Date. 20042021 AstraZeneca reports good progress of Covid-19 vaccine in trials.
1 min read AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety. 26032021 AstraZeneca COVID-19 vaccine trial data results in revised efficacy. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older.
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. For purportedly out-of-date data in the original phase 3 preliminary study AstraZeneca conducted a. 20072020 AstraZeneca and Oxford announced their partnership on the vaccine on April 30.
Added value of this study. 08122020 Topline data on the AstraZeneca vaccine from a Phase 3 trial were released in November. Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704.
A 10000-patient study testing the vaccine is being run in the United Kingdom and a separate 5000-patient test began. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing accuracy questions earlier this week surrounding a. The interim analysis published today in.
Results of Phase III trial. On 23 November 2020 the first interim data was released by Oxford University and AstraZeneca from the vaccines ongoing Phase III trials. The interim data reported a 70 efficacy based on combined results of 62 and 90 from different groups of participants who were given different dosages.
08032021 Data are expected soon in an early February interview AstraZeneca research chief Mene Pangalos had predicted results were four to six weeks away and could provide crucial answers to the remaining questions. 20072020 According to AstraZeneca late-stage trials of AZD1222 include a Phase III trial in Brazil 2000 participants ISRCTN89951424 a Phase IIbIII trial in. And Brazil and in a Phase 12 trial in South Africa.
The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. Another vaccine option in coming months. Investigators recently reported promising results in The Lancet.
Currently the vaccine candidate is being evaluated in Phase 23 trials in the UK. New vaccine efficacy results are reported now in The Lancet. We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa.
25032021 AstraZeneca on Thursday published an updated analysis confirming that a Phase 3 trial involving its COVID-19 vaccine was safe and effective and showed to. Estimated Study Completion Date.
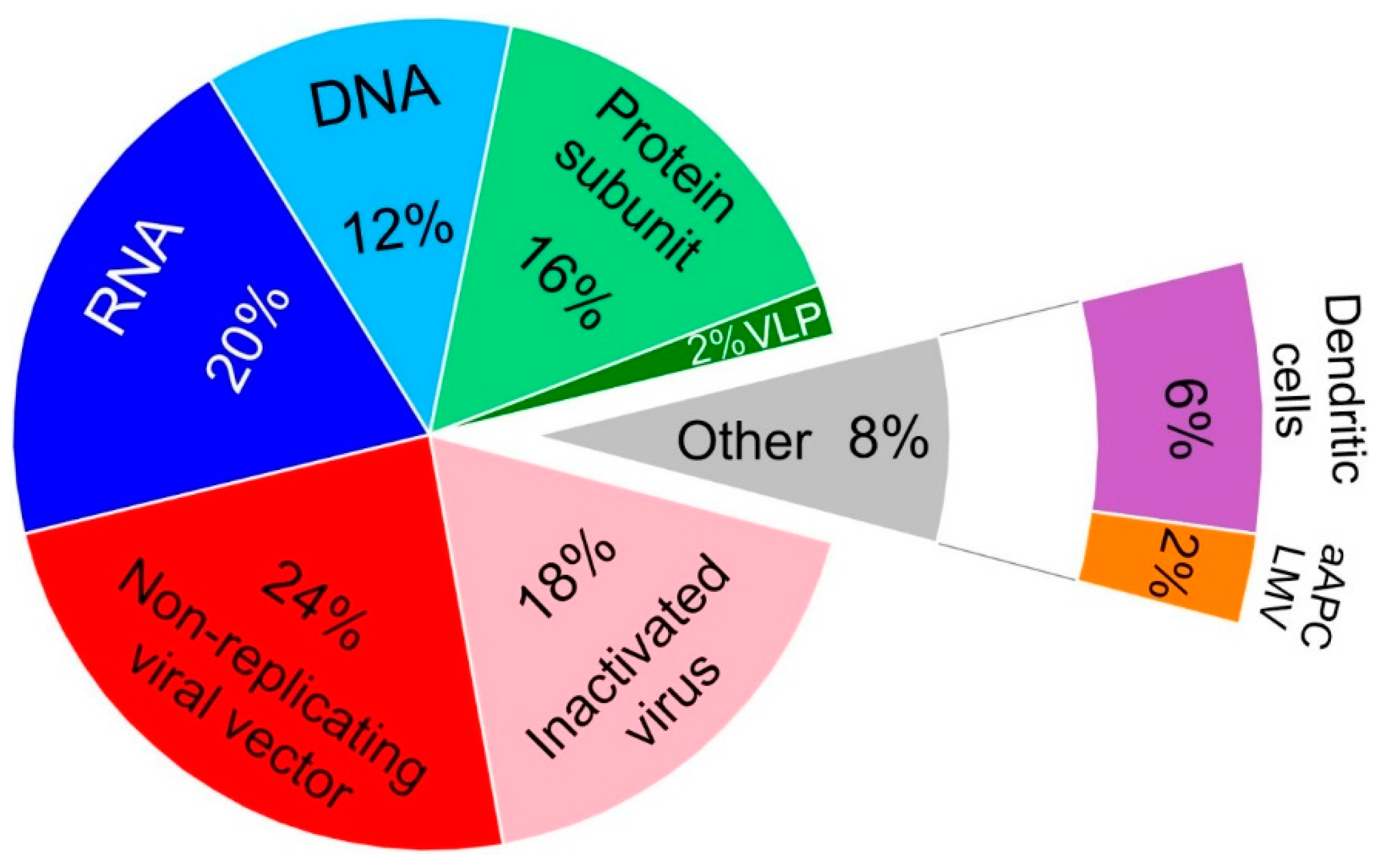
 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
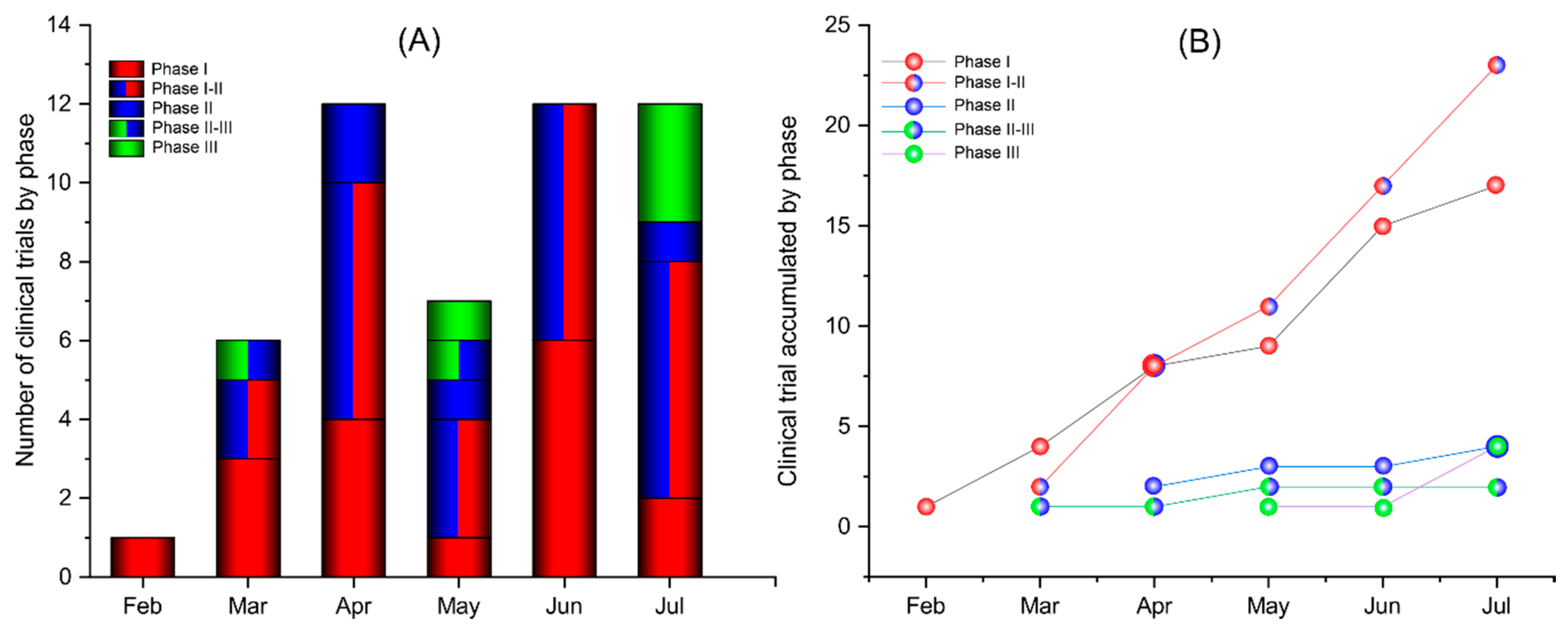
 Coronavirus Vaccine Updates From Astrazeneca To Moderna Here S Current Status Of Covid 19 Vaccines In Development
Coronavirus Vaccine Updates From Astrazeneca To Moderna Here S Current Status Of Covid 19 Vaccines In Development
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
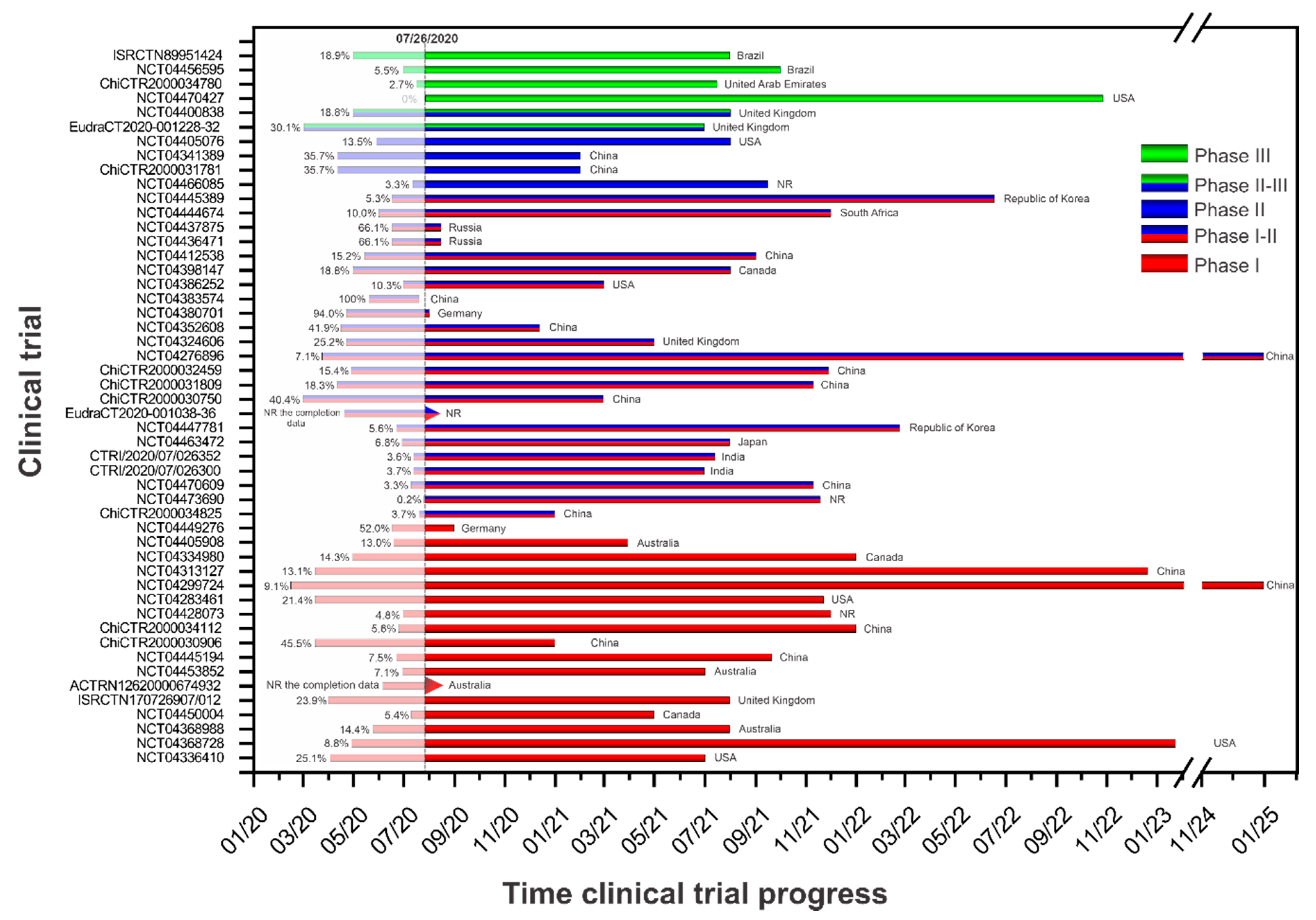 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
 How Soon Can We Expect Coronavirus Vaccine Crucial Data Expected By October End
How Soon Can We Expect Coronavirus Vaccine Crucial Data Expected By October End
 Phase 2 Trials Of Oxford Vaccine Could Begin At 3 4 Sites This Week India News The Indian Express
Phase 2 Trials Of Oxford Vaccine Could Begin At 3 4 Sites This Week India News The Indian Express
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times

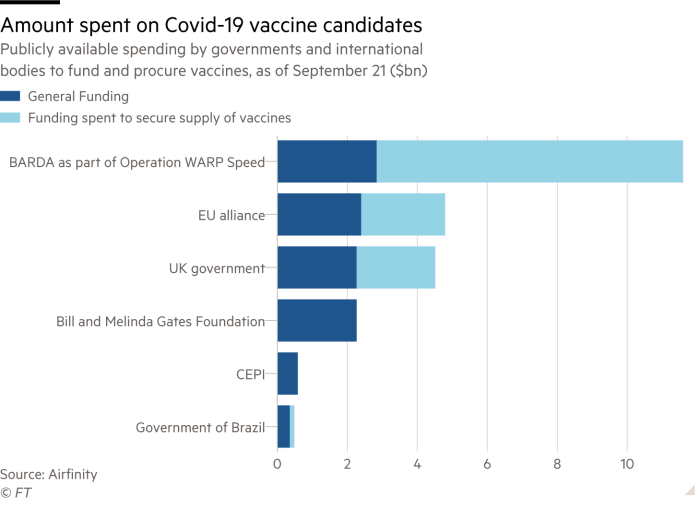 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
 Safety And Immunogenicity Of The Chadox1 Ncov 19 Vaccine Against Sars Cov 2 A Preliminary Report Of A Phase 1 2 Single Blind Randomised Controlled Trial The Lancet
Safety And Immunogenicity Of The Chadox1 Ncov 19 Vaccine Against Sars Cov 2 A Preliminary Report Of A Phase 1 2 Single Blind Randomised Controlled Trial The Lancet

 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
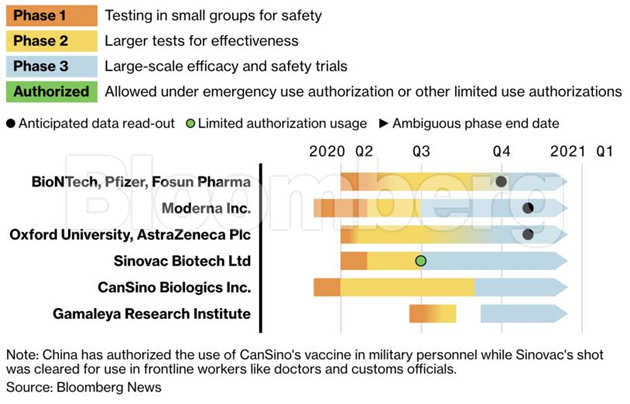
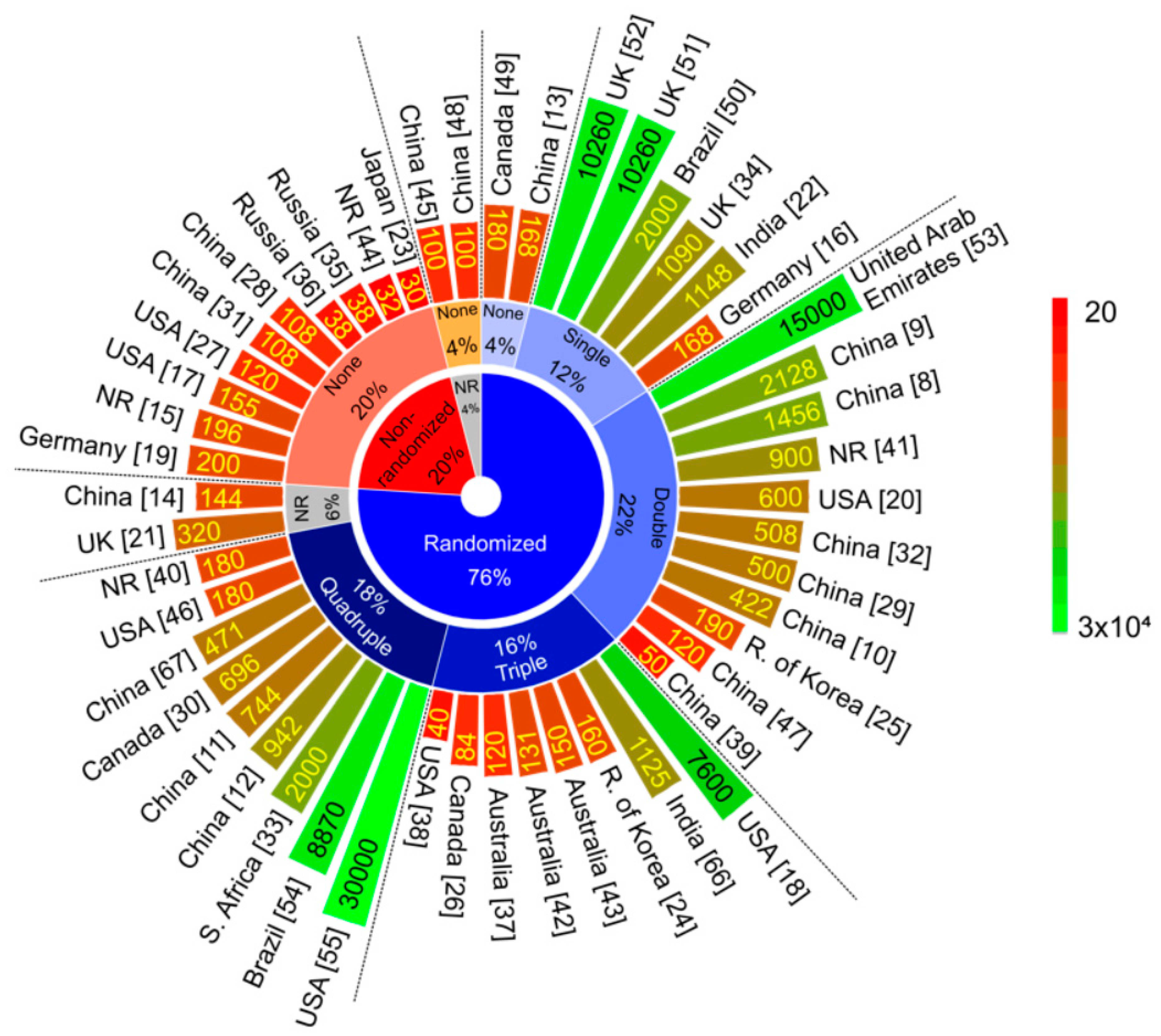 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Safety And Efficacy Of An Rad26 And Rad5 Vector Based Heterologous Prime Boost Covid 19 Vaccine An Interim Analysis Of A Randomised Controlled Phase 3 Trial In Russia The Lancet
Safety And Efficacy Of An Rad26 And Rad5 Vector Based Heterologous Prime Boost Covid 19 Vaccine An Interim Analysis Of A Randomised Controlled Phase 3 Trial In Russia The Lancet
Post a Comment for "Astrazeneca Vaccine Phase 3 Results Date"