Astrazeneca Phase 3 Results Usa
11092020 The first 5 trials likely to have phase 3 results are rounded out by Germanys BNTPfizer vaccine and the NIHModerna vaccine from the US. 25102020 First announcement of safety results from a phase 3 trial.
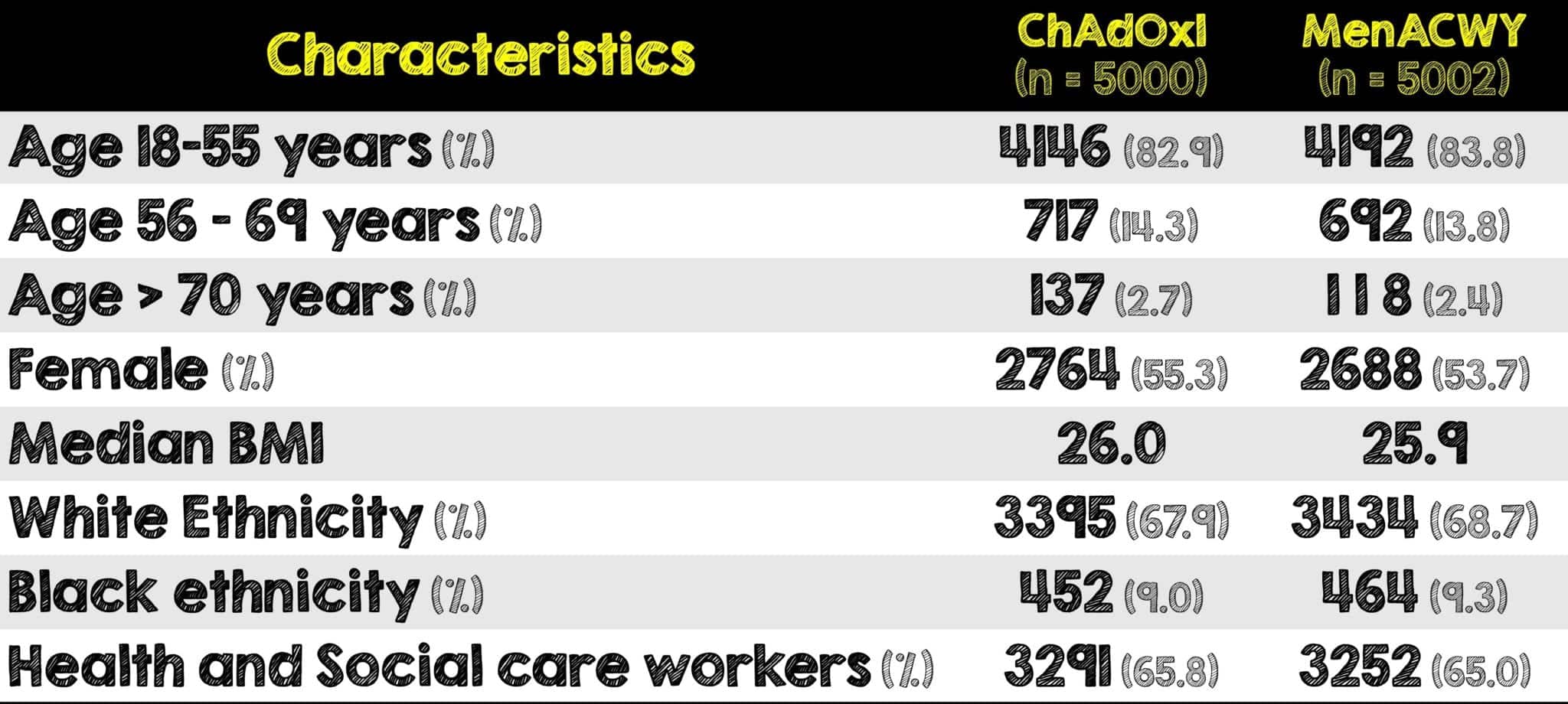 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Partnered with one of the worlds leading contract research organizations CROs called IQVIA AstraZeneca has launched the Phase 3 double-blind placebo-controlled study of AZD1222 for the possible prevention of COVID-19 in adults.

Astrazeneca phase 3 results usa. Phase 3 Trial of Vaccine Candidate Against RSV Lower Respiratory Tract Infections Meets Primary Endpoint. ChAdOx1 nCoV-19 has an acceptable safety profile and has been found to be efficacious against symptomatic COVID-19 in this interim analysis of ongoing clinical trials. Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation.
23102020 Clinical trials for the AstraZeneca Oxford coronavirus vaccine AZD1222 have resumed across the world with regulators in the US UK Brazil South Africa and Japan confirming that it was safe to do so. 25032021 Positive high-level results from the primary analysis of the Phase III trial of AZD1222 in the US have confirmed vaccine efficacy consistent with the pre-specified interim analysis announced on Monday 22 March 2021. The interim analysis for efficacy was based on 11636 participants accruing 131.
CoronaVac Theres no publication for this. Results demonstrated vaccine efficacy of 76 CI. In its third-quarter results announcement.
05022021 LONDON Reuters - AstraZeneca expects to have results from a US. But it was an announcement by the head of the public research institute running the trial. Phase 3 clinical trial of its COVID-19 vaccine candidate AZD1222.
AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US. Currently the vaccine candidate is being evaluated in Phase 23 trials in the UK. Based on the RML data a Phase 1 trial of the candidate vaccine began on April 23 in healthy volunteers in the UK.
Both are for 30000 participants. The Food and Drug Administration FDA today authorised the restart in the US following the resumption of trials in other countries in recent. 22032021 The US Phase III trial called D8110C00001 was led by AstraZeneca and funded by the Biomedical Advanced Research and Development Authority BARDA part of the office of the Assistant Secretary for Preparedness and Response ASPR at the US Department of Health and Human Services HHS in collaboration with the Department of Defense Joint Program Executive.
AstraZeneca COVID-19 vaccine becomes 3rd in United States to begin Phase 3 trials. The interim analysis published today in. They both started phase 3 trials in the US on the same day at the end of July although the BNTPfizer one is a compressed phase 123 trial.
07092018 SOURCE is a Phase III multicentre randomised double-blinded parallel-group placebo-controlled trial for 48 weeks in adult patients with severe asthma who require continuous treatment with ICS plus long-acting beta2-agonists LABA and chronic treatment with maintenance oral corticosteroid OCS therapy. And Brazil and in a Phase 12 trial in South Africa. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
These results have been presented to the independent Data Safety Monitoring Board. The results are also consistent with the vaccines earlier results and in line with what would be expected from an inactivated virus vaccine. 03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose.
Another vaccine option in. This study literally just started last week according to US. 11 February 2021 904 am.
Clinical trial of its COVID-19 vaccine in the next four to six weeks the. About AstraZeneca in Respiratory Disease. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector.
05112020 Pharmaceutical company AstraZeneca said Wednesday that phase three trial results for its COVID-19 vaccine could be ready later this year. 59 to 86 after a first. AstraZeneca has begun a US.
Investigators recently reported promising results in The Lancet. The Biomedical Advanced Research and Development Authority.
 Usa Chile And Peru Interim Trial Data Show Oxford Astrazeneca Vaccine Is Safe And Highly Effective University Of Oxford
Usa Chile And Peru Interim Trial Data Show Oxford Astrazeneca Vaccine Is Safe And Highly Effective University Of Oxford
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Coronavirus Vaccine The Story Of The Race To Solve The Covid Conundrum Euronews
Coronavirus Vaccine The Story Of The Race To Solve The Covid Conundrum Euronews
 Covid Vaccine Astrazeneca Phase Iii Trials Interim Results Youtube
Covid Vaccine Astrazeneca Phase Iii Trials Interim Results Youtube
 Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
 U K Authorizes Oxford Astrazeneca Coronavirus Vaccine The Washington Post
U K Authorizes Oxford Astrazeneca Coronavirus Vaccine The Washington Post
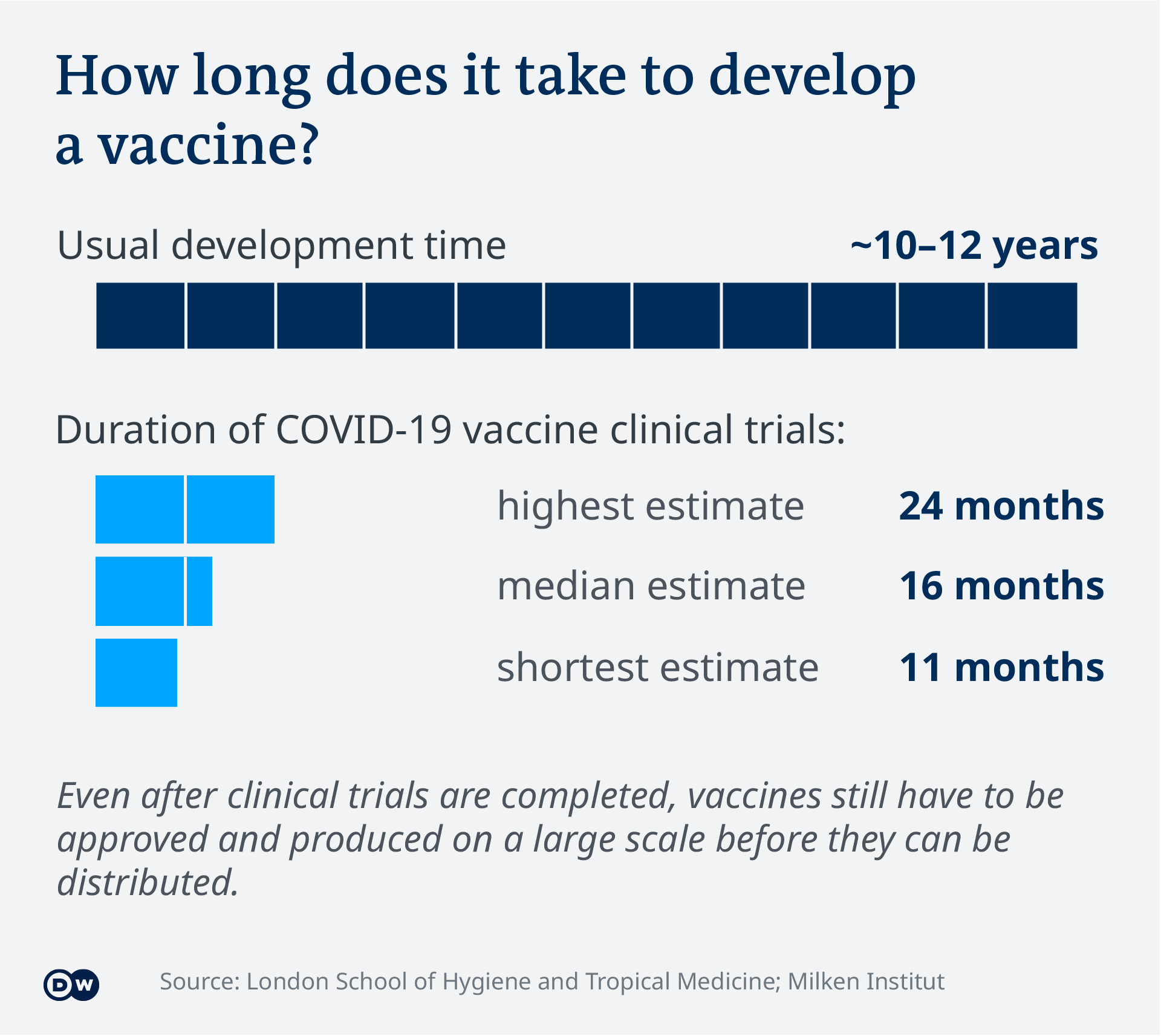 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 The Lead Covid 19 Vaccine Candidates Where We Are And What You Should Know Lexology
The Lead Covid 19 Vaccine Candidates Where We Are And What You Should Know Lexology
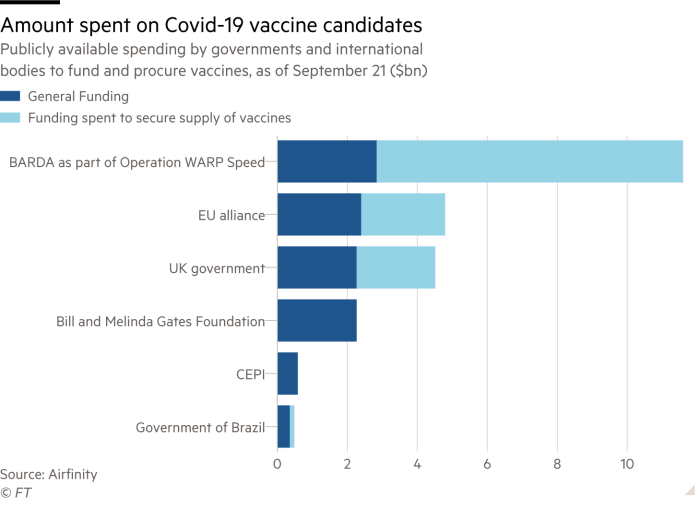 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Searching For A Vaccine Chemviews Magazine Chemistryviews
Searching For A Vaccine Chemviews Magazine Chemistryviews
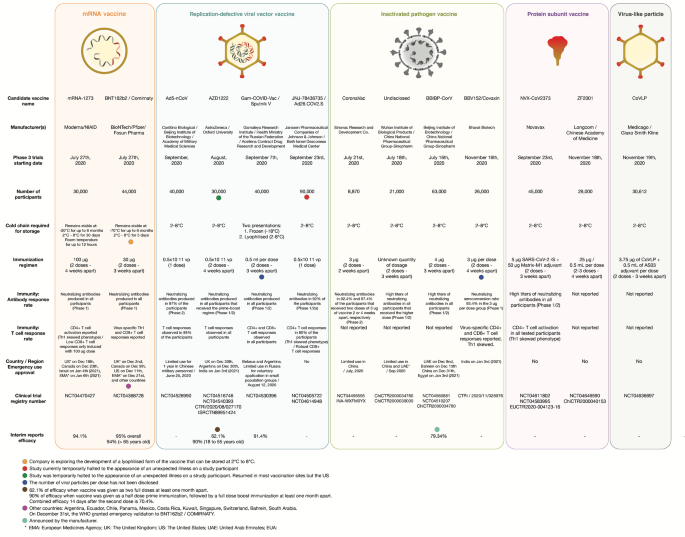 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Astrazeneca Says Shot 90 Effective
Astrazeneca Says Shot 90 Effective
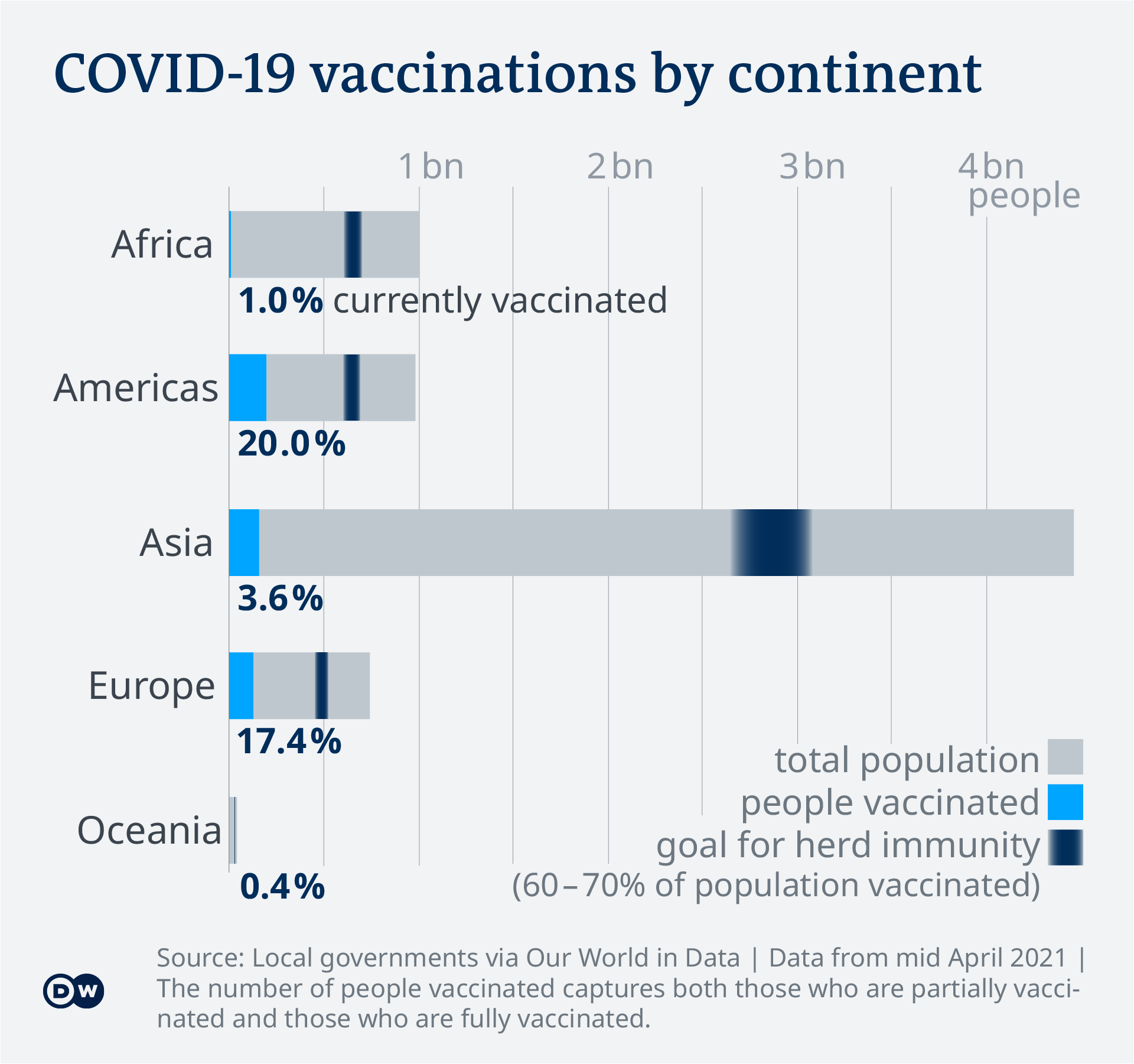 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
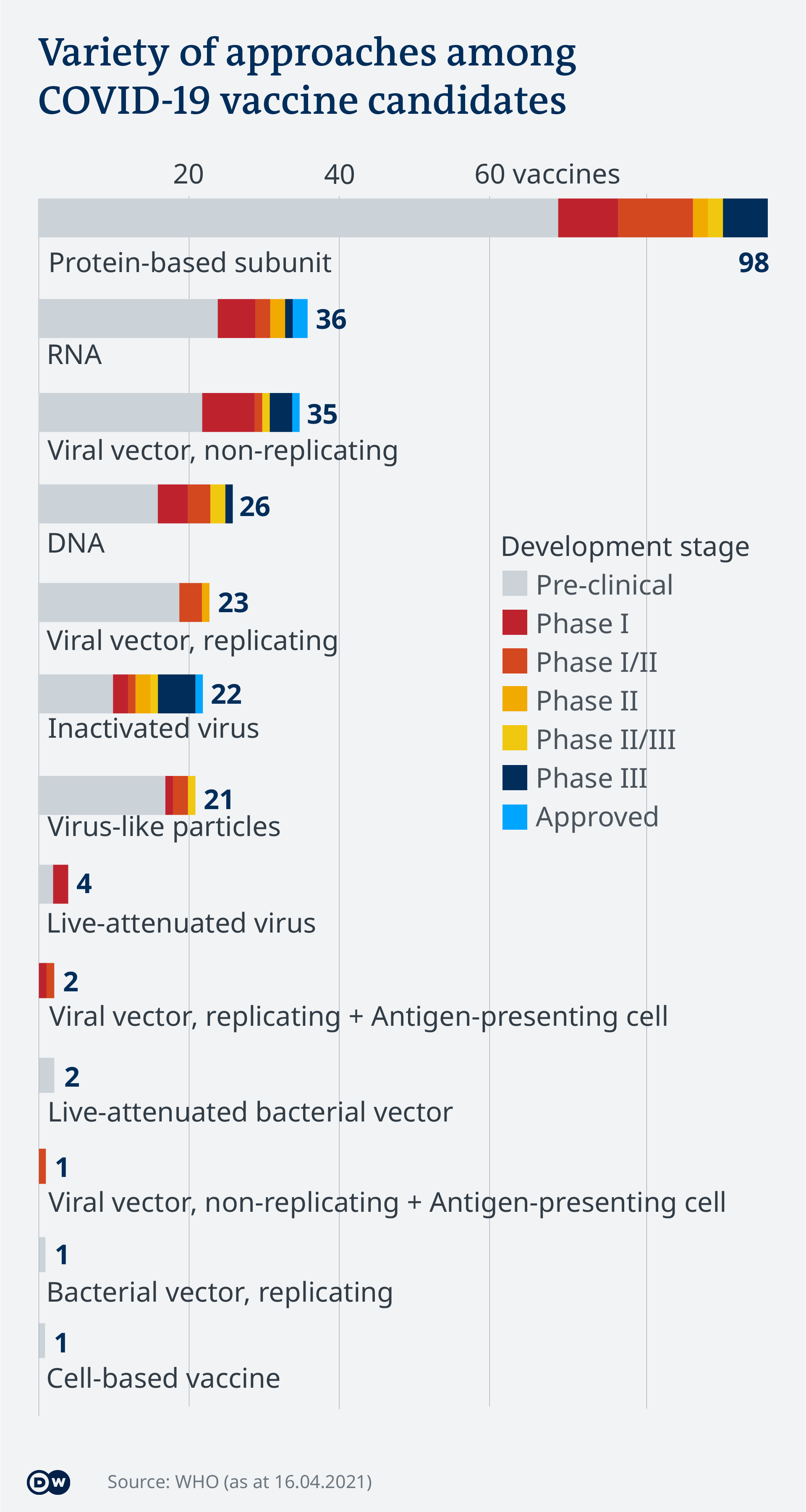 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Astrazeneca Covid Vaccine 79 Effective In U S Trial
Astrazeneca Covid Vaccine 79 Effective In U S Trial
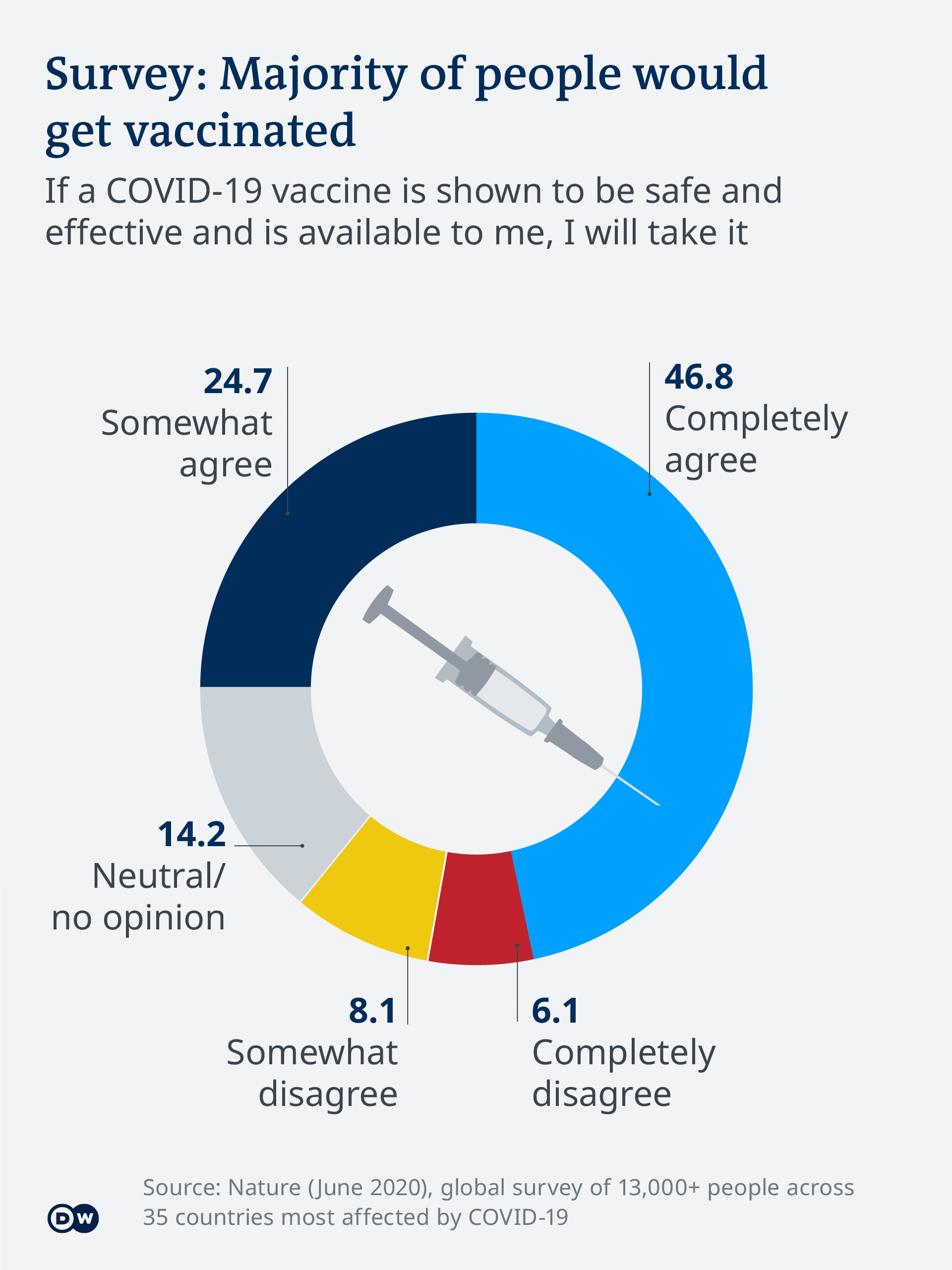 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Post a Comment for "Astrazeneca Phase 3 Results Usa"