Astrazeneca Phase 3 Results Covid
British-Swedish pharmaceutical major AstraZeneca on Monday 22 March said its vaccine demonstrated efficacy of 79 per cent at preventing symptomatic Covid-19 and 100. 11 February 2021 904 am.
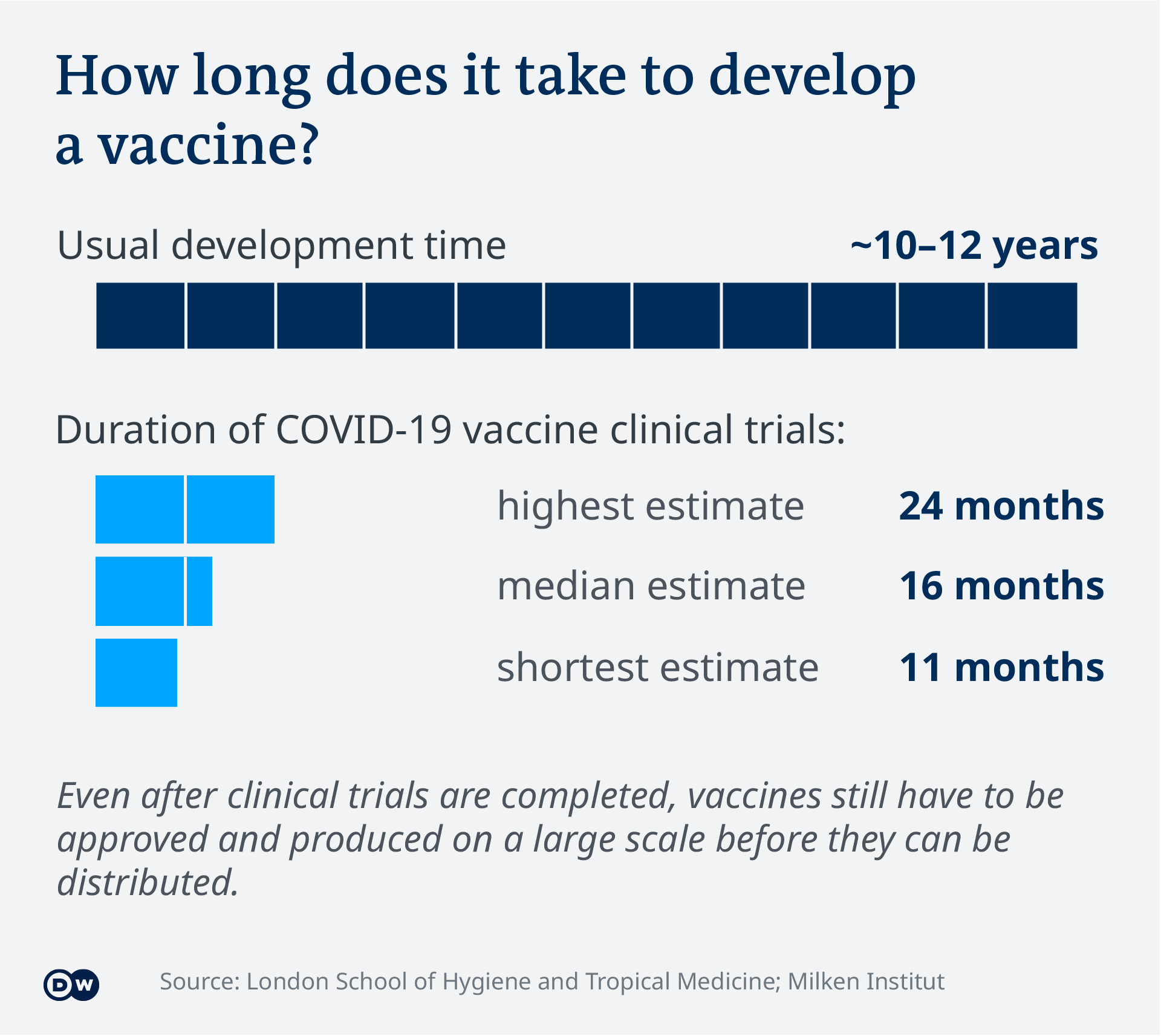 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
31 it has begun enrolling up to 30000 people in a phase 3 trial for its experimental COVID-19 vaccine.

Astrazeneca phase 3 results covid. AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing Covid-19 more than 22 days after the first dose. The company now says. The results come after some countries in Europe briefly suspended the vaccine over fears of harmful side effects.
The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19. AstraZeneca anticipates having Phase 3 trial results by the end of March potentially giving the US. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature.
The University of Rochester Medical Center URMC has joined a phase 3 clinical trial for a potential coronavirus vaccine being developed by AstraZeneca and the University of Oxford known as. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US.
A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704. New vaccine efficacy results are reported now in The Lancet.
04022021 AstraZenecas Covid-19 vaccine proves safe and effective in Phase III trial. 22032021 AstraZeneca COVID Vaccine Shows 79 Efficacy in US Phase-3 Trial. 20072020 These results together with the induction of both humoral and cellular immune responses support large-scale evaluation of this candidate vaccine in an ongoing phase 3 program the researchers.
Another vaccine option in coming monthsBut in order to do so the company needs to smooth over its delivery delays with current commitments. 20042021 Two months later on March 3 the company announced an early analysis of the trial results finding that Covaxin has an efficacy of 806 percent. The trial funded by the.
We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa. Another vaccine option in. 05112020 Pharmaceutical company AstraZeneca said Wednesday that phase three trial results for its COVID-19 vaccine could be ready later this year.
25032021 AstraZeneca issues new Phase 3 data for COVID vaccine confirming safety and efficacy. AstraZeneca said Wednesday that a primary analysis of its Phase 3. 22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and.
Of the first 43 volunteers to get Covid-19 only 7. CEO Pascal Soriot said during an earnings call Thursday the company is ramping up its production of vaccines in coming months targeting. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US.
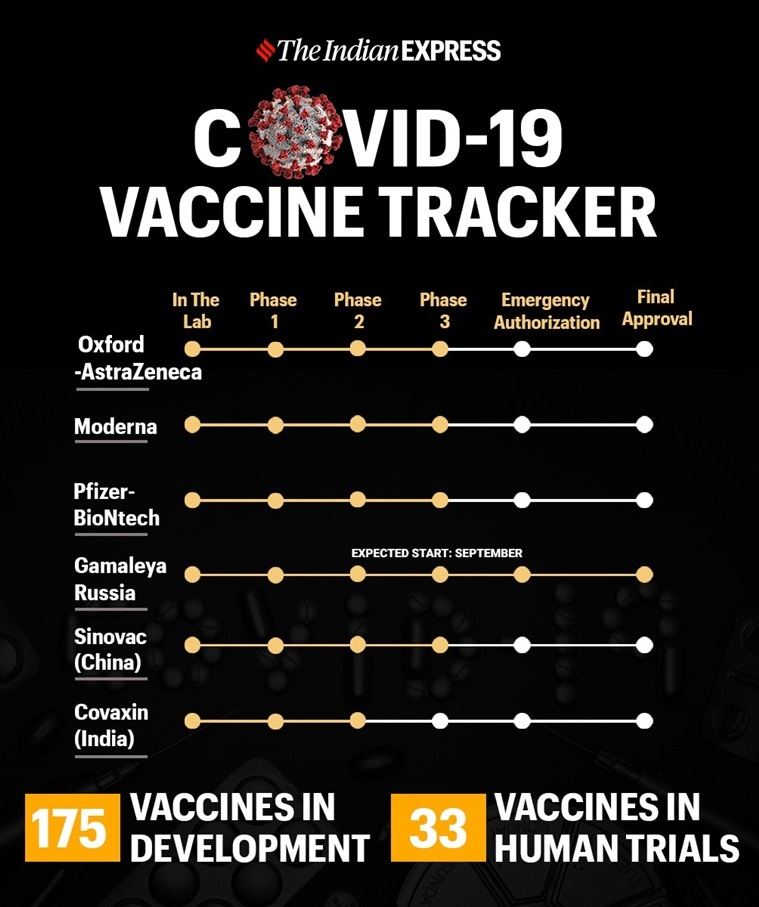 Coronavirus Covid 19 Vaccine Status Latest Update Oxford University Pfizer Moderna Russia Bharat Biotech Covaxin Status Check
Coronavirus Covid 19 Vaccine Status Latest Update Oxford University Pfizer Moderna Russia Bharat Biotech Covaxin Status Check

 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
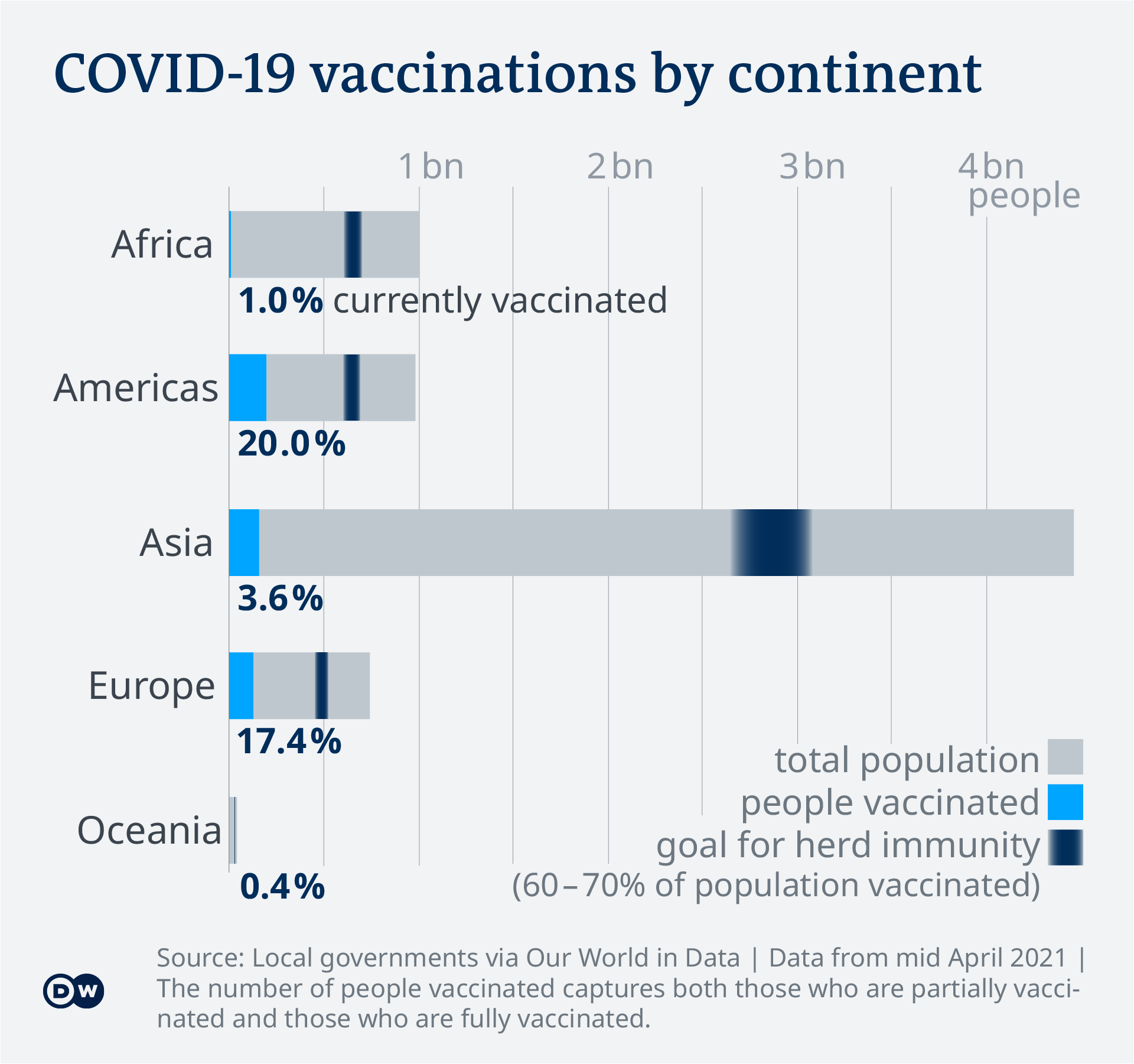 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
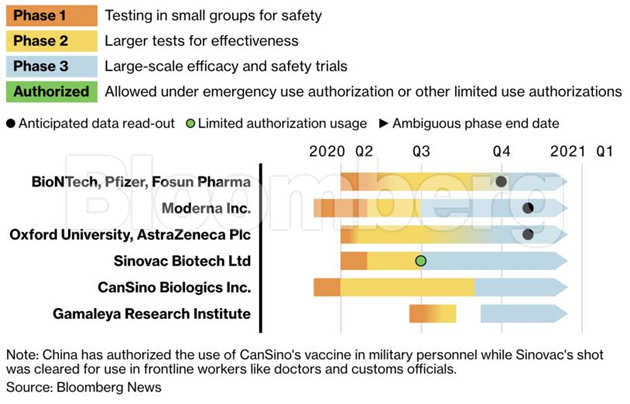
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
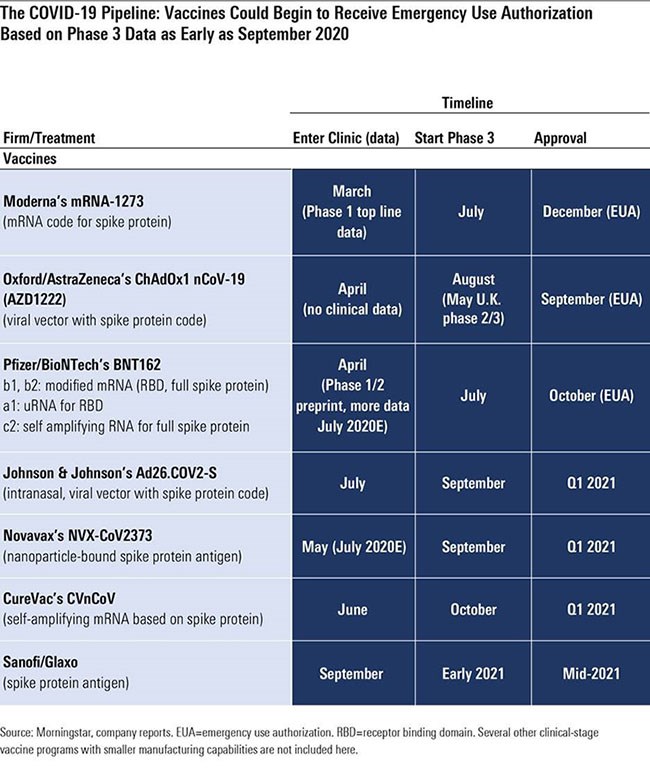 What A Covid 19 Vaccine Means For Investors Morningstar
What A Covid 19 Vaccine Means For Investors Morningstar

 Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
 Astrazeneca S Covid 19 Vaccine Proves Safe And Effective In Phase Iii Trial
Astrazeneca S Covid 19 Vaccine Proves Safe And Effective In Phase Iii Trial
 Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
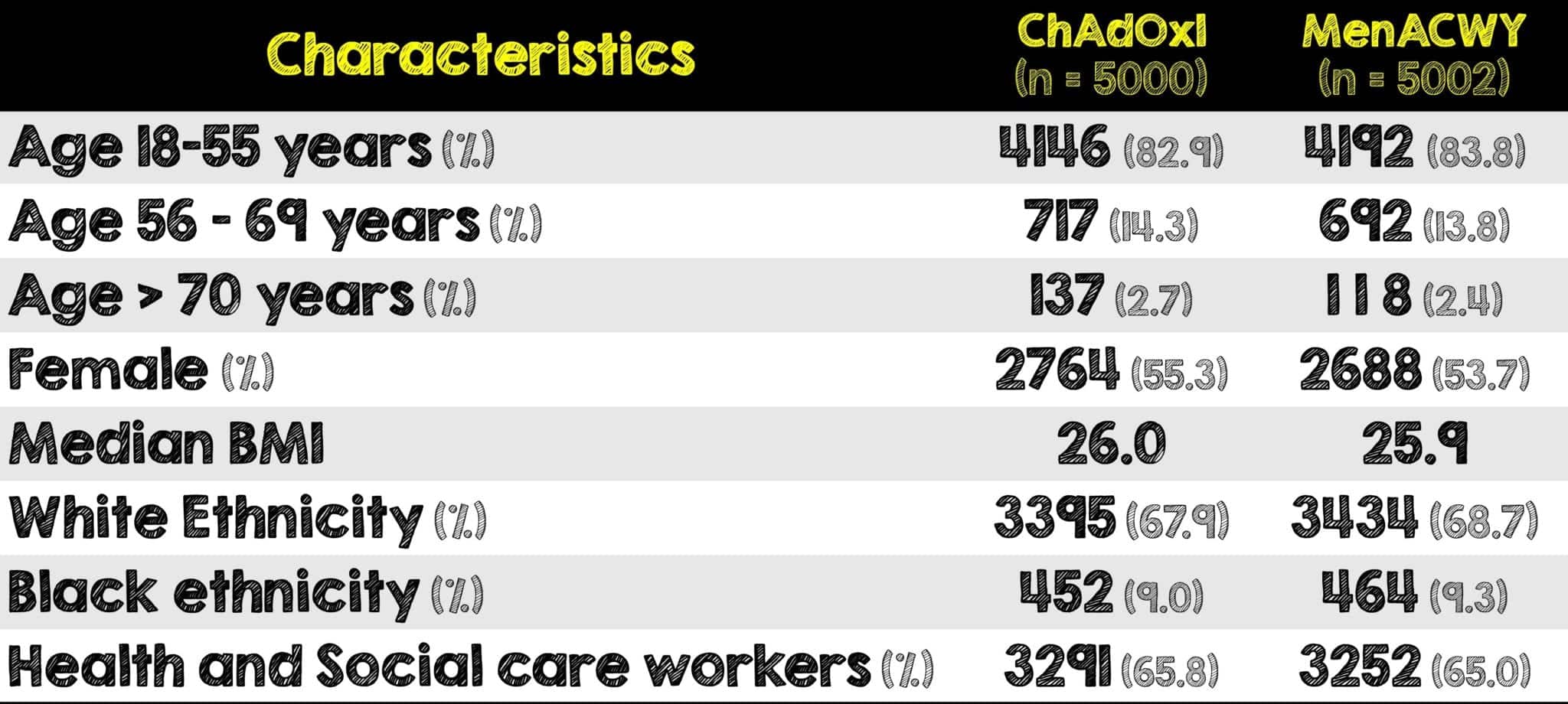 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
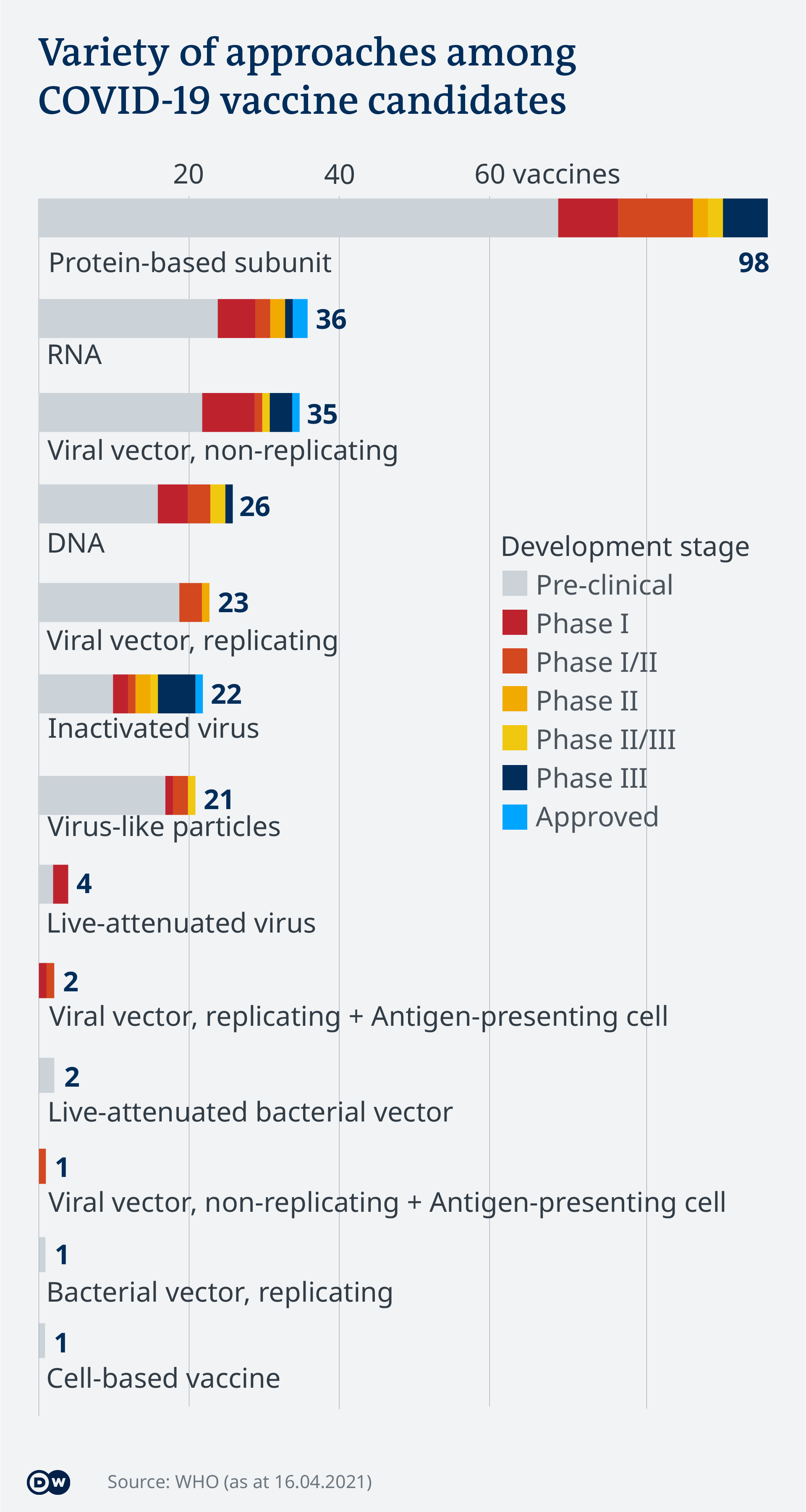 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Oxford Astrazeneca S Phase Iii Covid 19 Vaccine Data Could Be Available By December Biospace
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
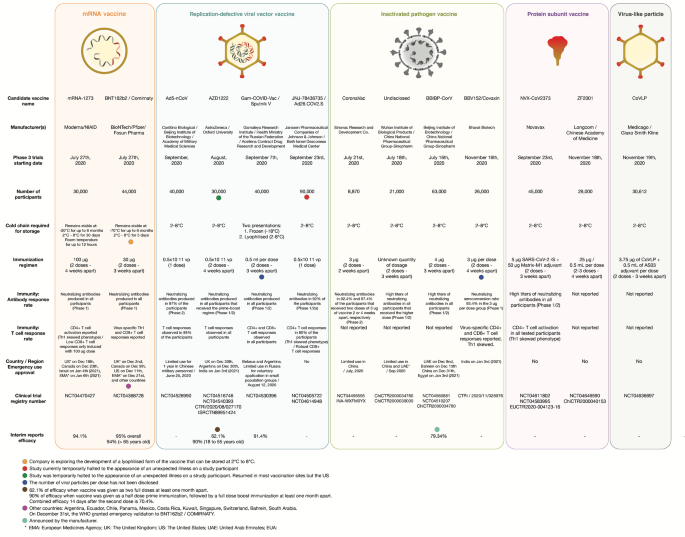 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Post a Comment for "Astrazeneca Phase 3 Results Covid"