Astrazeneca Phase 3 Results Us
At one point the US. 03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose.
 Latest U S Will Share Its Astrazeneca Vaccine Supply With The World
Latest U S Will Share Its Astrazeneca Vaccine Supply With The World
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector.

Astrazeneca phase 3 results us. 09042021 In an email an AstraZeneca spokesperson declined to respond specifically to Faucis statements or the advisers concerns but pointed CNN to AstraZenecas Phase 3. Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it. Hinges on forthcoming results from a large Phase 3 study run in the country and in South America.
These results have been presented to the independent Data Safety Monitoring Board. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19. 25032021 AstraZeneca announced today its results from the phase 3 clinical trial that it carried out in the US Chile and Peru.
28102020 October 28 2020 - AstraZeneca recently announced that the Phase 3 clinical trial for its COVID-19 vaccine AZD122 has resumed in the US after FDA deemed it safe to do so. This was from a planned interim analysis from a phase 3 study that was run in the US and South America. 22032021 The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79 at preventing symptomatic COVID-19 and 100 efficacy at preventing severe disease and hospitalisation.
All clinical trials for AZD122 have now resumed globally after regulators in the UK Brazil South Africa and Japan also reviewed all safety data from trials globally and. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. 12032021 Meantime regulators in the United States have been waiting for new AstraZeneca data expected in the next few weeks from a Phase 3.
First on 22 March 2021 US NIH and AstraZeneca both issued press releases reported 79 efficacy against symptomatic COVID-19. This is a new trial required for approval of the vaccine for use in the US in consequence of the companys amateurish mismanagement of its original clinical trial. 25032021 Positive high-level results from the primary analysis of the Phase III trial of AZD1222 in the US have confirmed vaccine efficacy consistent with the pre-specified interim analysis announced on Monday 22 March 2021.
Clinical trial of its COVID-19 vaccine in the next four to six weeks the. 20042021 On March 22 the University of Oxford and the British-Swedish company AstraZeneca became the fourth team to deliver results from a large-scale Phase 3 trial that could lead the FDA. Results demonstrated vaccine efficacy of 76 CI.
The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US. 22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and.
WHO warns against vaccine nationalism AstraZeneca is nearing the end of. 05022021 LONDON Reuters - AstraZeneca expects to have results from a US. Had bet more on AstraZenecas vaccine than any other and even with three alternatives available could still benefit from its availability.
AstraZeneca expects Phase 3 results soon. 08032021 While the vaccine appears to meet the FDAs standards for authorization its future in the US.

 Astrazeneca Says Shot 90 Effective
Astrazeneca Says Shot 90 Effective
 Astrazeneca S Covid 19 Vaccine Is 79 Percent Effective In U S Trial Science News
Astrazeneca S Covid 19 Vaccine Is 79 Percent Effective In U S Trial Science News
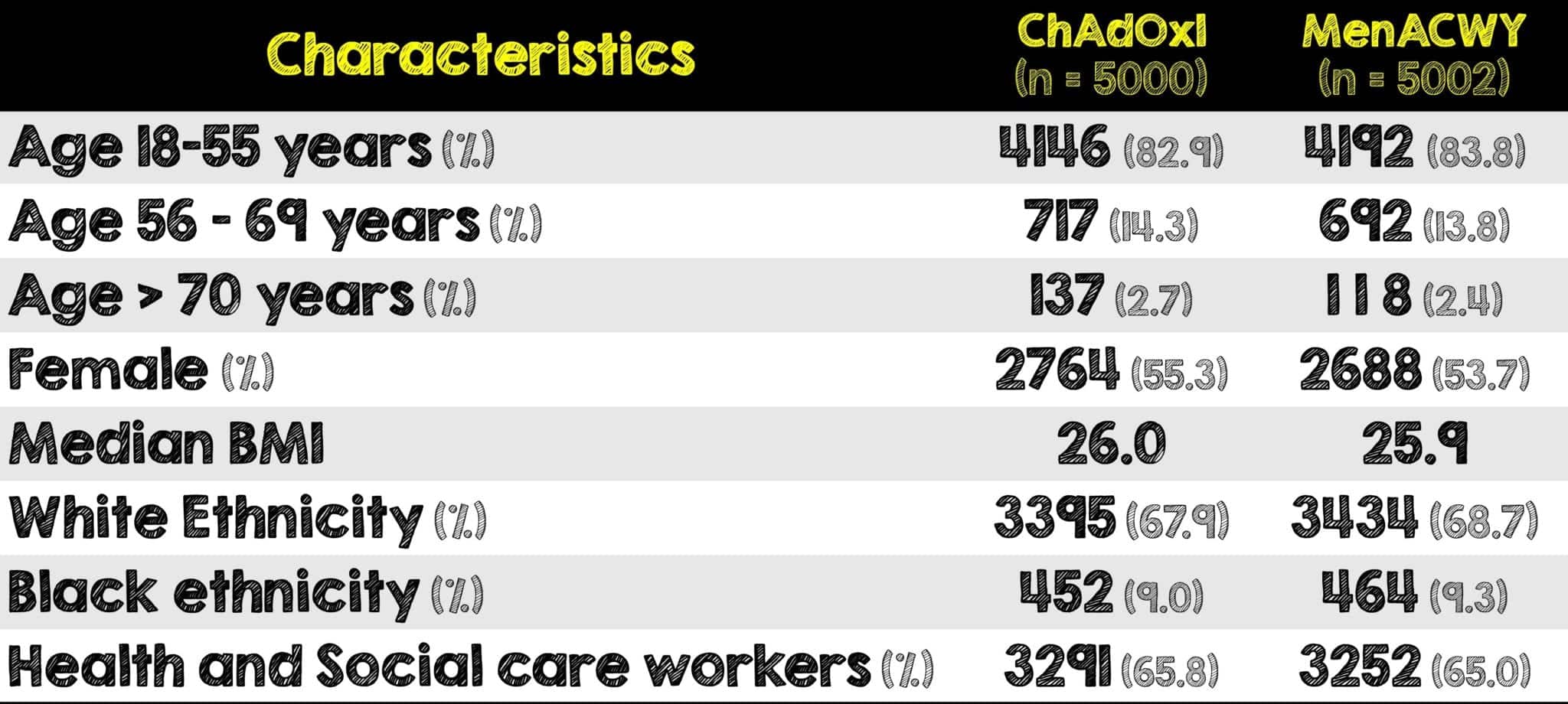 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
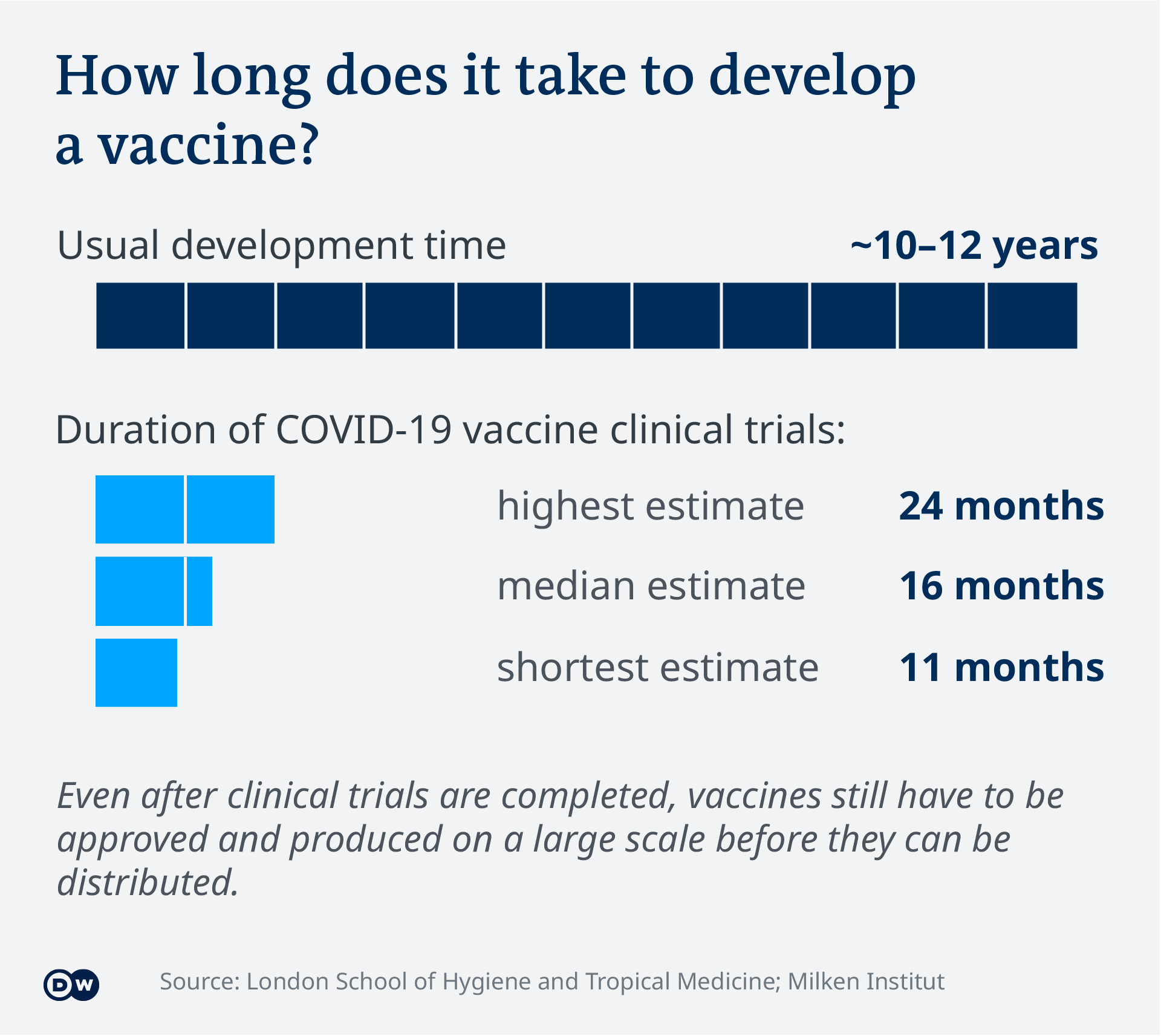 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Astrazeneca Resumes Its Covid 19 Vaccine Trials In The U K Coronavirus Updates Npr
Astrazeneca Resumes Its Covid 19 Vaccine Trials In The U K Coronavirus Updates Npr
 Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
 Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
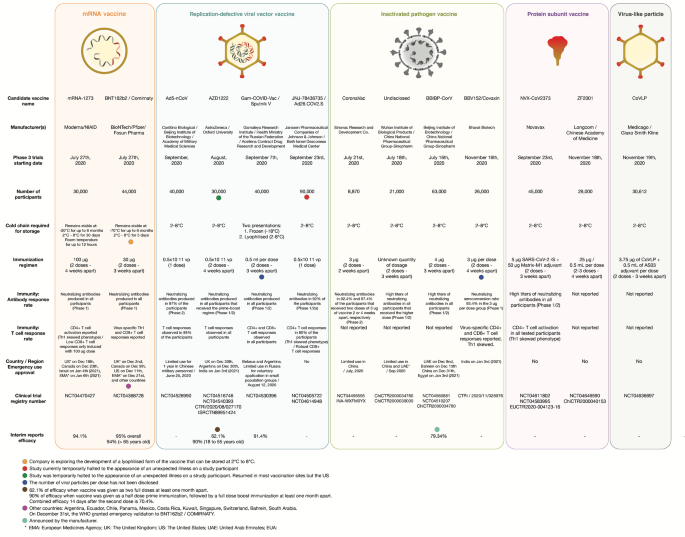 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
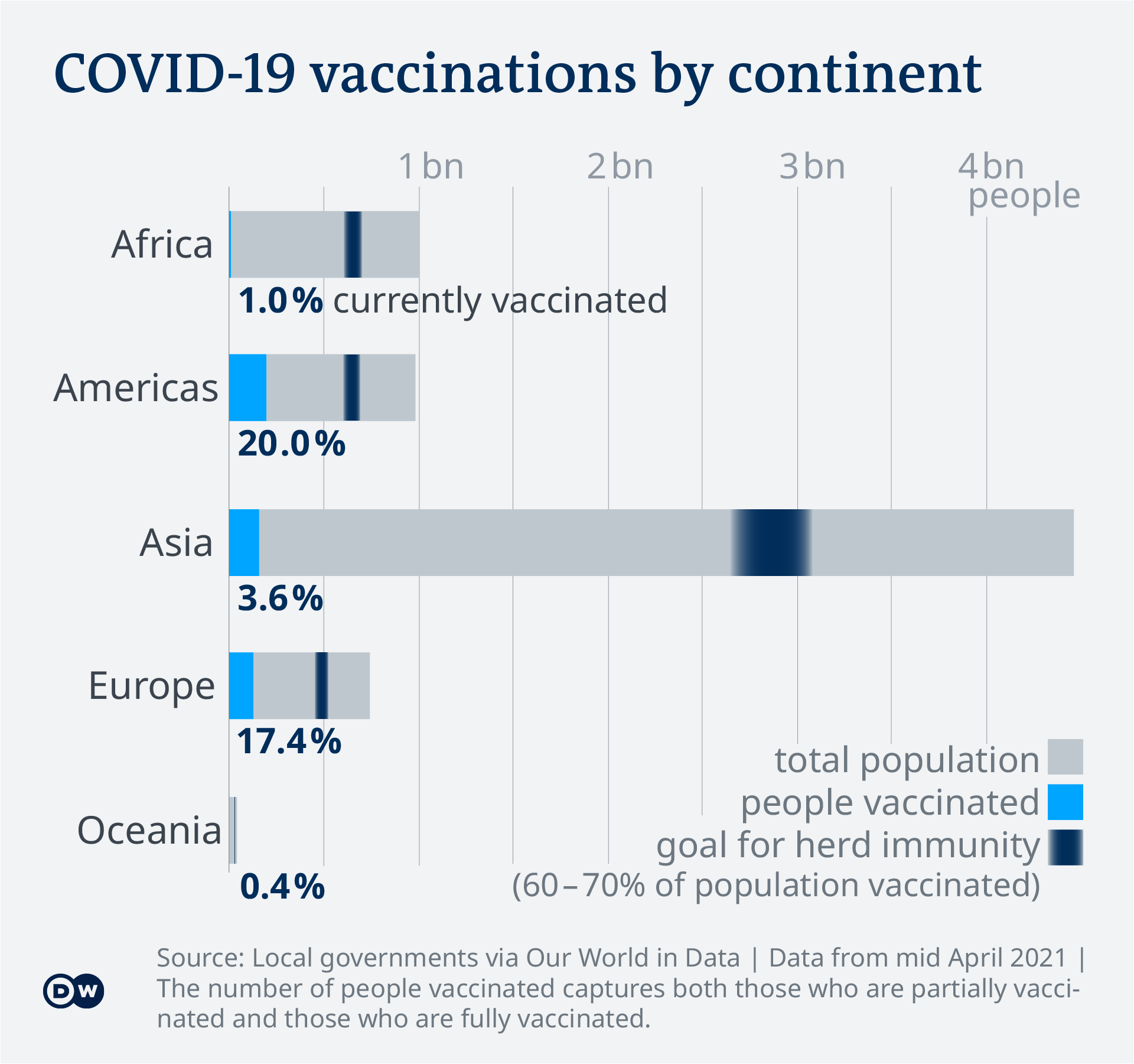 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Us Study Of Astrazeneca Vaccine Proves Efficacy Video Abc News
Us Study Of Astrazeneca Vaccine Proves Efficacy Video Abc News
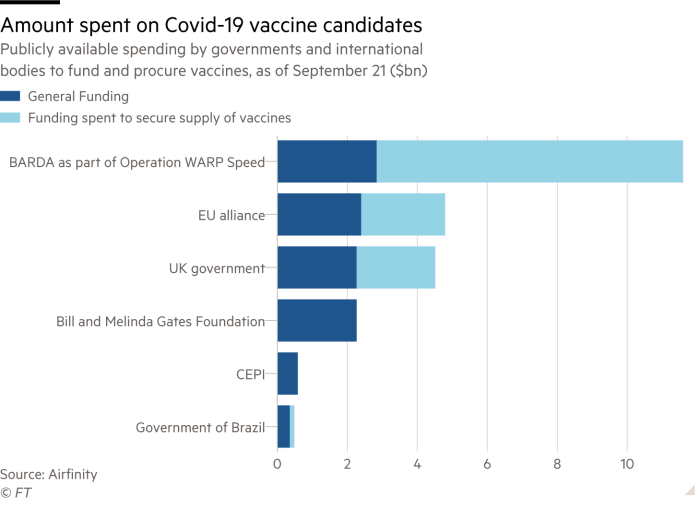 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
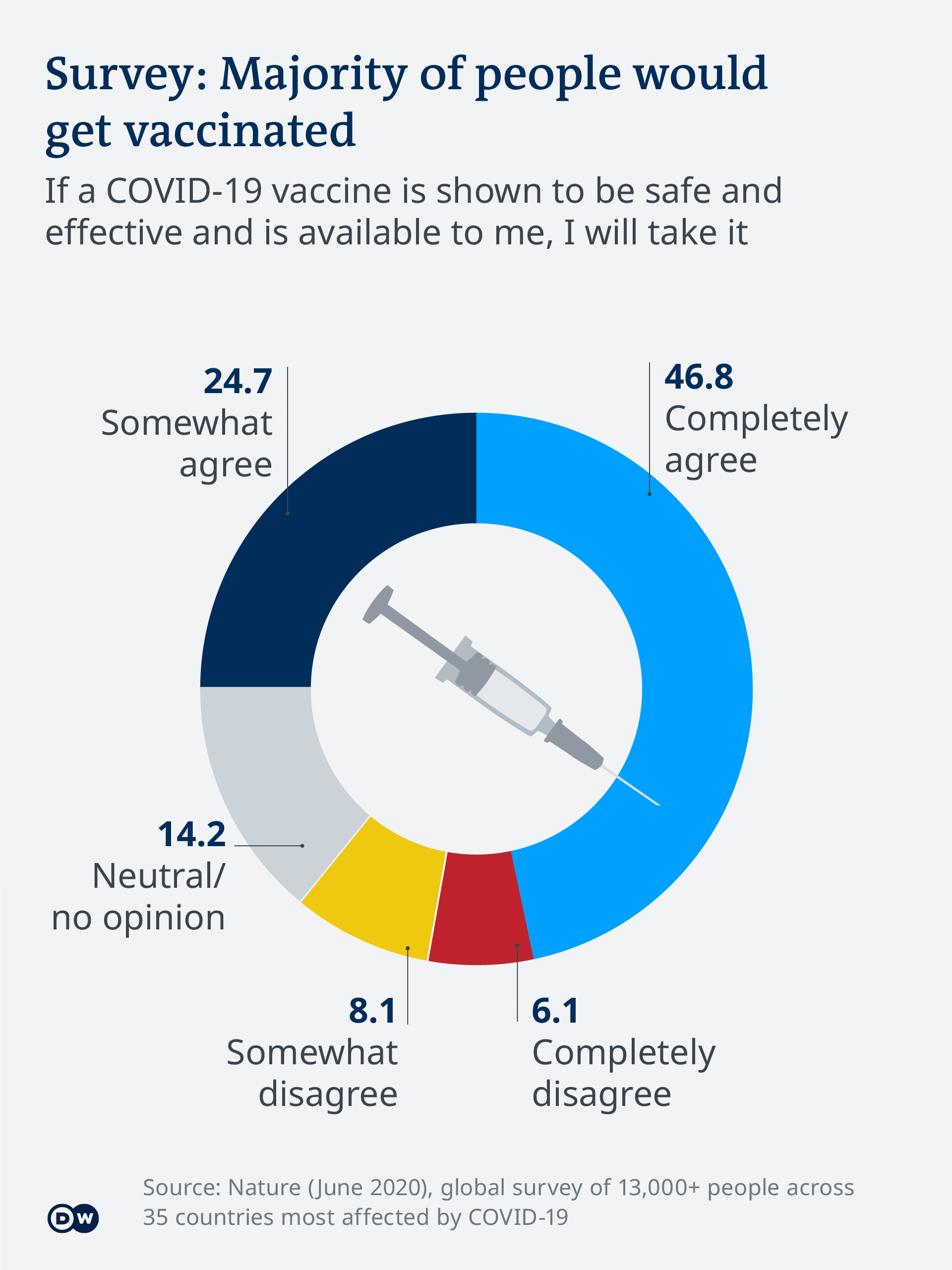 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
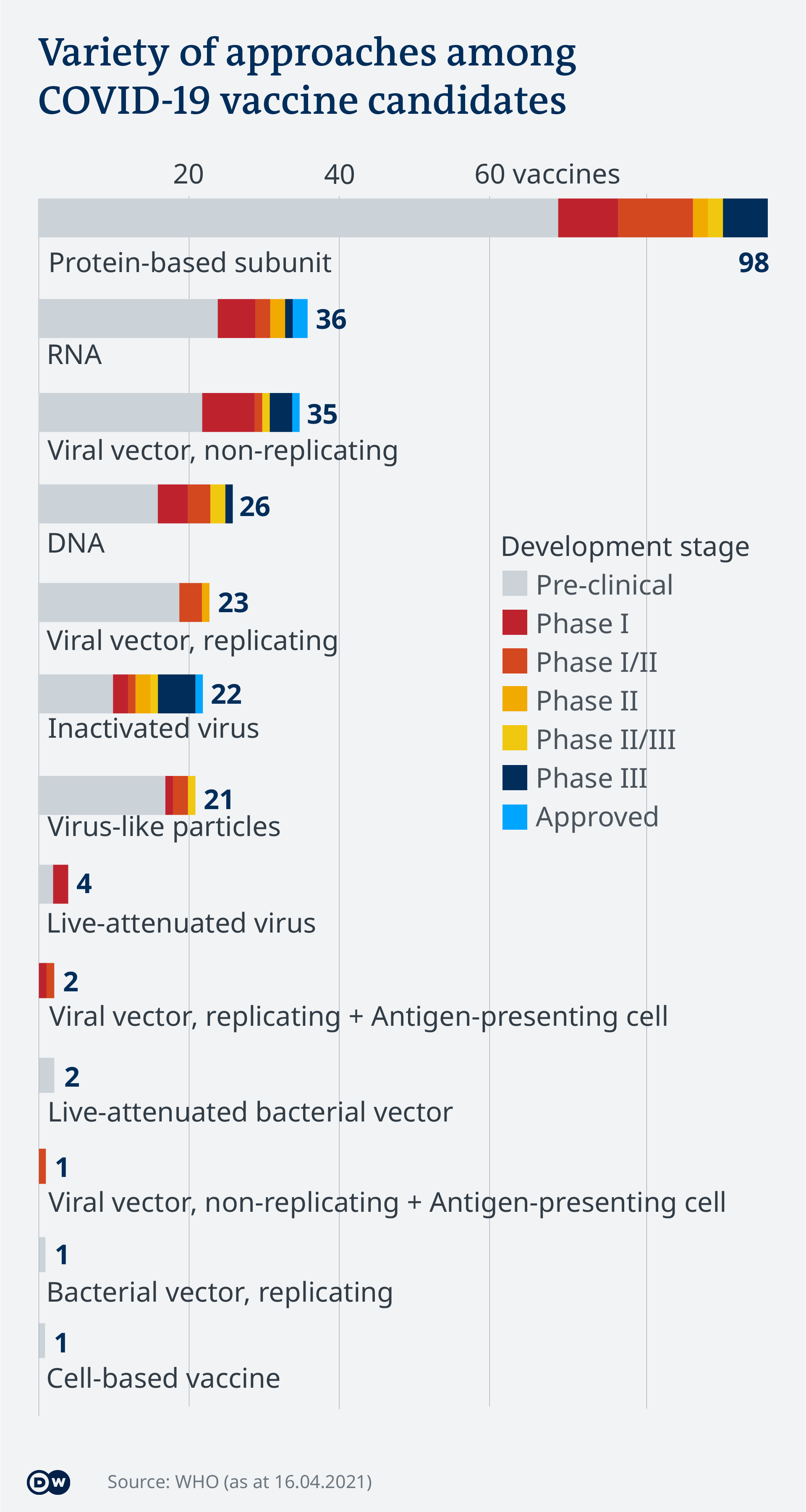 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
Post a Comment for "Astrazeneca Phase 3 Results Us"