Astrazeneca Phase 3 Results Date
Here we present the safety and immunogenicity results of a phase 2 component of a phase 23 multicentre study using ChAdOx1 nCoV-19 at two different doses in adults including those aged 5669 years and 70 years and older and in a one-dose or two-dose regimen. But the company noted results were much better 90 for participants who had mistakenly received a half then full dose of vaccine rather than two full doses 62.
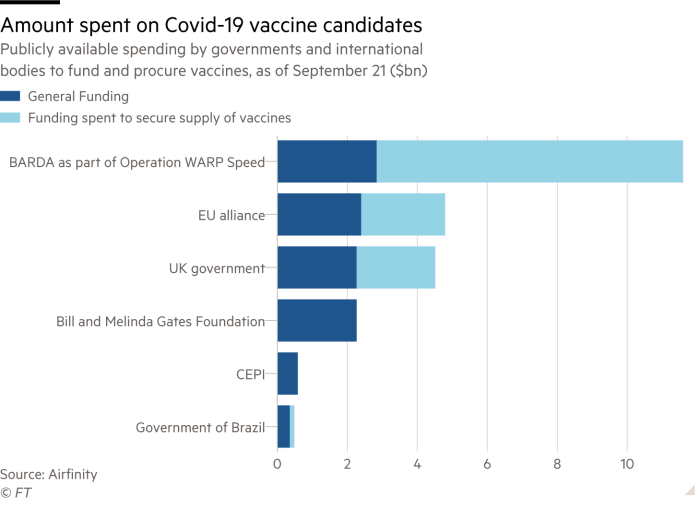 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19.

Astrazeneca phase 3 results date. Currently the vaccine candidate is being evaluated in Phase 23 trials in the UK. 3-min read AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US. 03022021 COVID-19 Vaccine AstraZeneca confirms 100 protection against severe disease hospitalisation and death in the primary analysis of Phase III trials PUBLISHED 3 February 2021 3 February 2021 0700 GMT.
ChAdOx1 nCoV-19 has an acceptable safety profile and has been found to be efficacious against symptomatic COVID-19 in this interim analysis of ongoing clinical trials. In the case of the prominent Moderna mRNA-1273 trial the start date of the Phase 3 study was July 27 2020. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing accuracy questions earlier this week surrounding a.
But what about the promising BNT162 trial. AstraZeneca COVID Vaccine 79 Efficacious in Phase 3 Interim Results - Medscape - Mar 22 2021. And Brazil and in a Phase 12 trial in South Africa.
Actual Study Start Date. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. This interim safety and efficacy analysis was based on 32449 participants accruing 141 symptomatic cases of COVID-19.
Based on the RML data a Phase 1 trial of the candidate vaccine began on April 23 in healthy volunteers in the UK. 11 February 2021 904 am. Scopus 380 Google Scholar.
Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. The interim analysis published today in. Investigators recently reported promising results in The Lancet.
New vaccine efficacy results are reported now in The Lancet. 25032021 Positive high-level results from the primary analysis of the Phase III trial of AZD1222 in the US have confirmed vaccine efficacy consistent with the pre-specified interim analysis announced on Monday 22 March 2021. 22032021 The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79 at preventing symptomatic COVID-19 and 100 efficacy at preventing severe disease and hospitalisation.
Another vaccine option in coming months. 09042021 Results from the Phase 3 PREVENT-19 study for Novavax Covid-19 vaccine candidate NVX-CoV2373 are expected shortly and positive data could pave the way for the company to submit an Emergency. 08032021 When AstraZeneca first reported data in November it said a two-shot regimen was on average 70 effective at preventing COVID-19.
On March 22 the University of Oxford and the British-Swedish company AstraZeneca became the fourth team to deliver results from a large-scale Phase 3 trial that could lead the FDA. Actual Primary Completion Date. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704.
Study found it was 79 effective against symptomatic COVID-19. Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation. Now the estimated primary completion date is actually October 27 2021 a little over a year from the start date.
And the estimated final end date is the same. 22032021 The statement from NIAID comes a day after AstraZeneca said the interim results of their phase III US. The interim analysis for efficacy was based on.
These results have been presented to the independent Data Safety Monitoring Board. Estimated Study Completion Date.
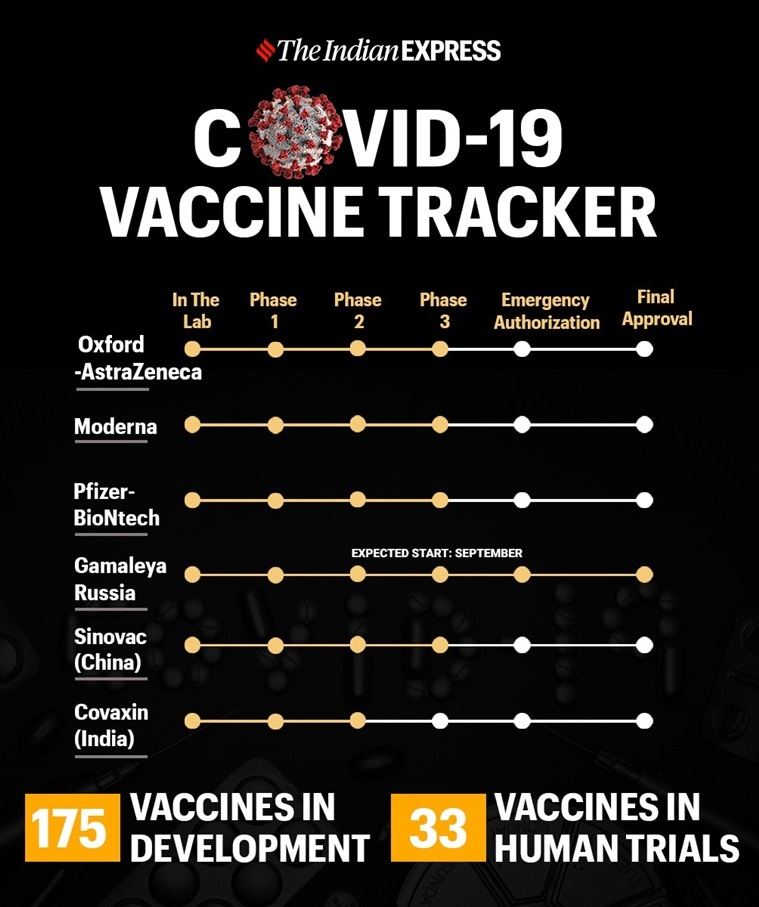 Coronavirus Covid 19 Vaccine Status Latest Update Oxford University Pfizer Moderna Russia Bharat Biotech Covaxin Status Check
Coronavirus Covid 19 Vaccine Status Latest Update Oxford University Pfizer Moderna Russia Bharat Biotech Covaxin Status Check
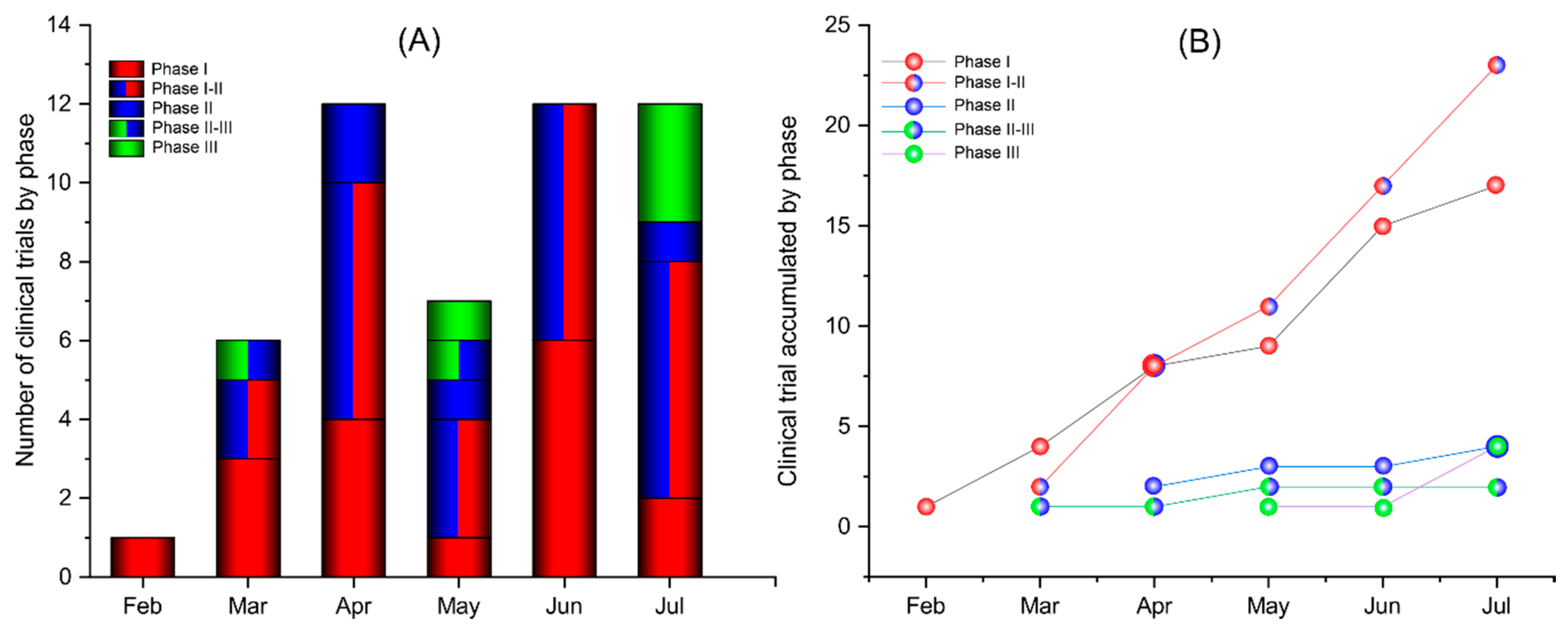 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results

 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials

 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
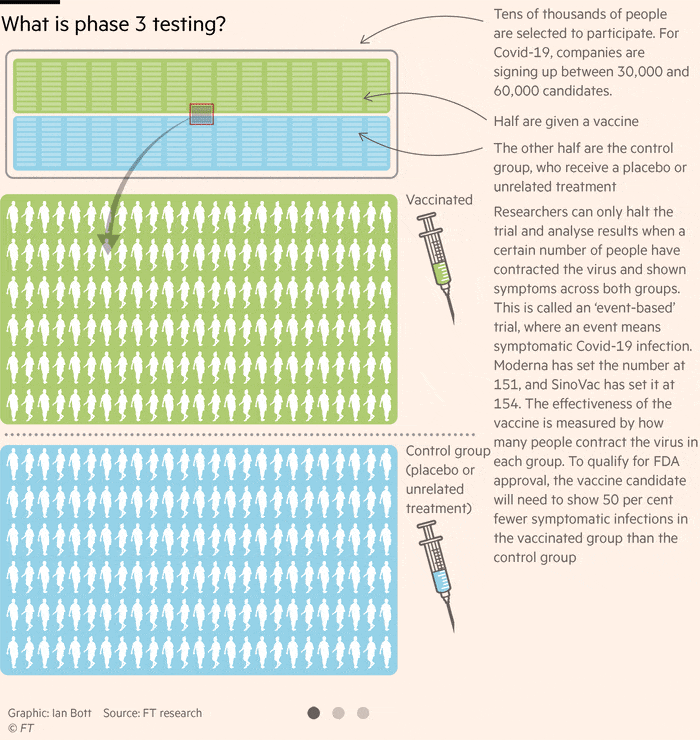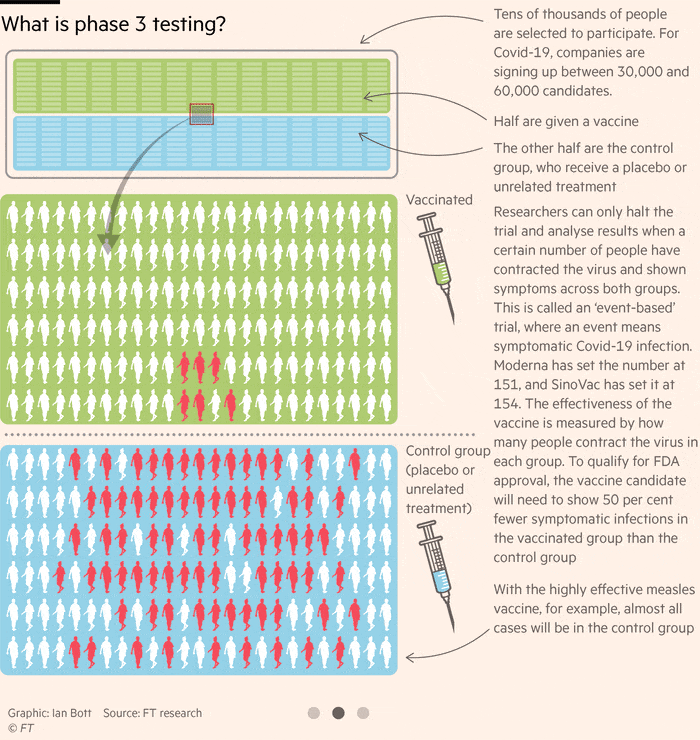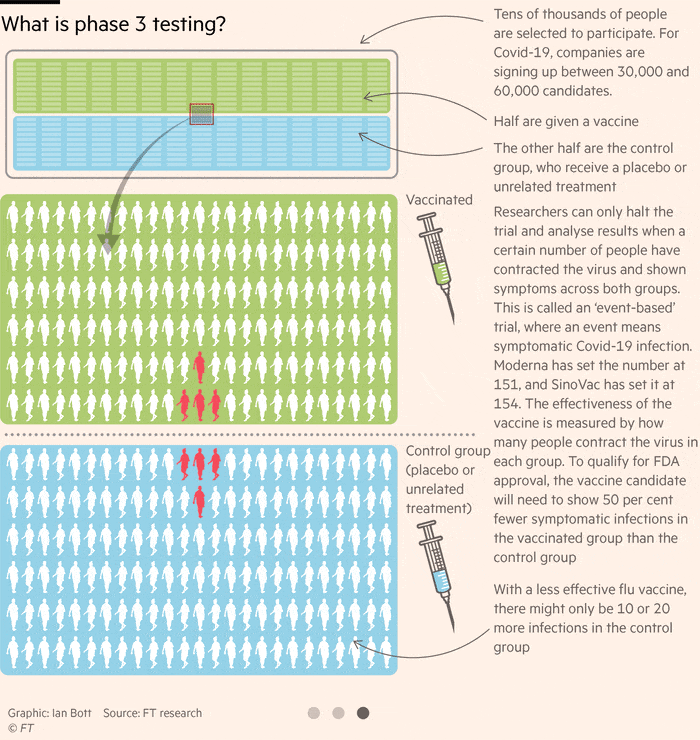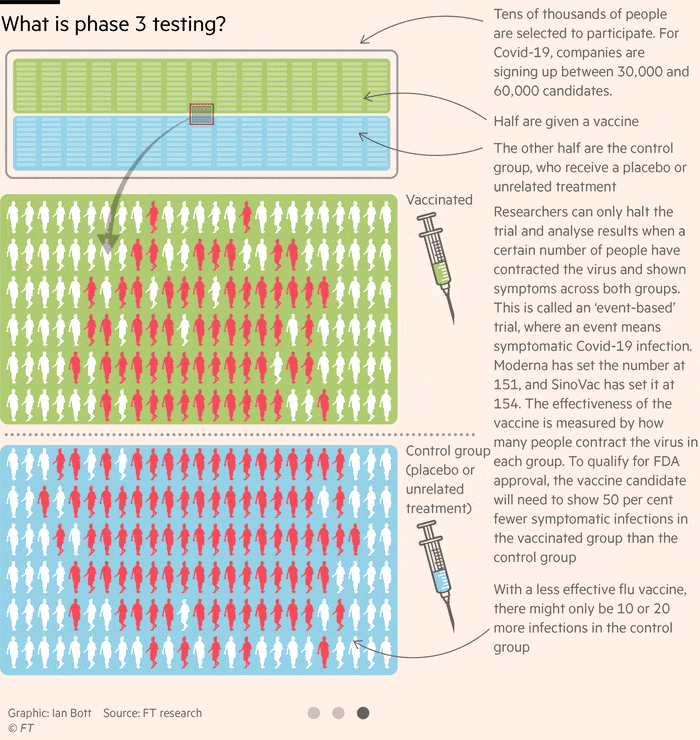 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
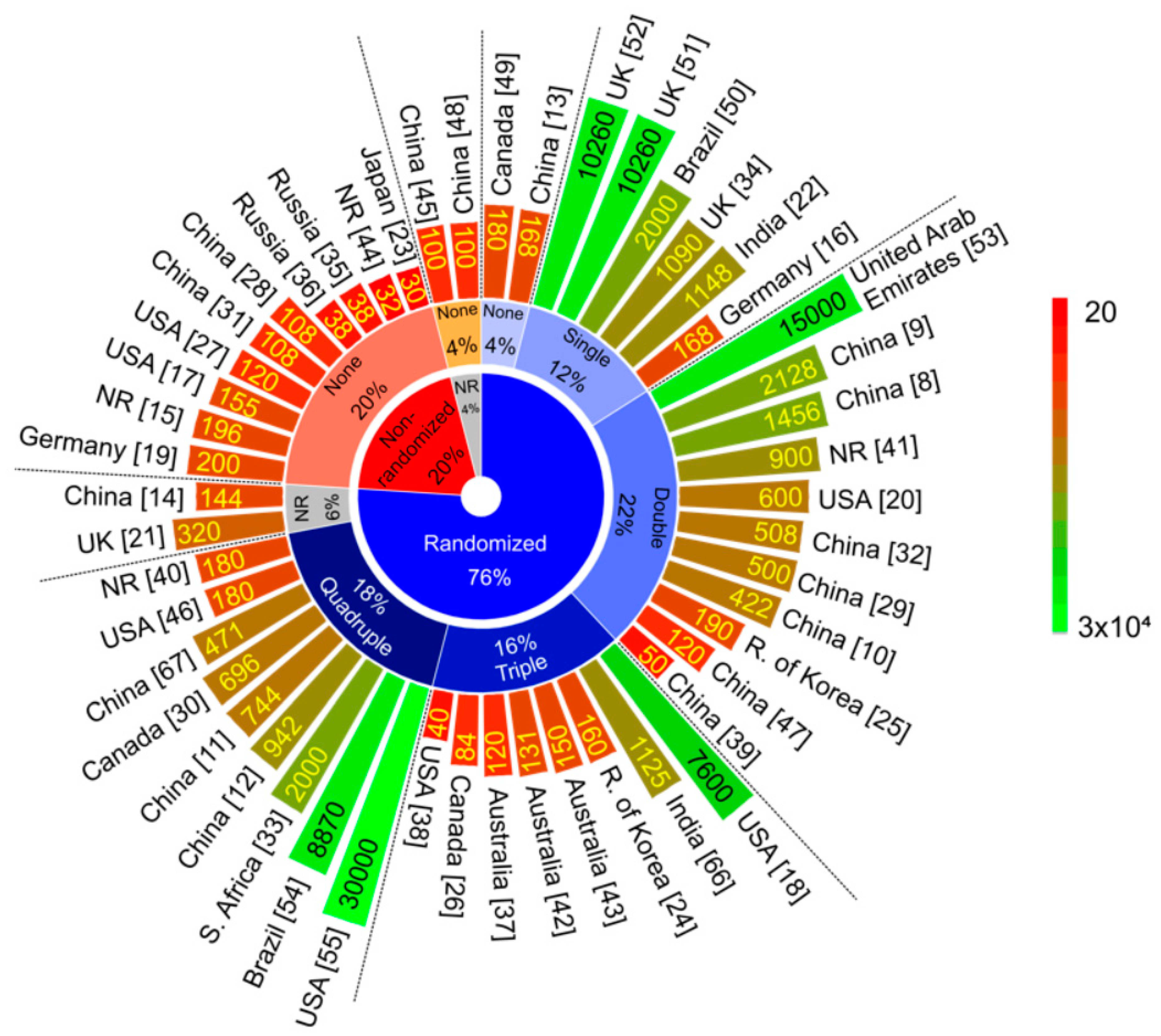 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
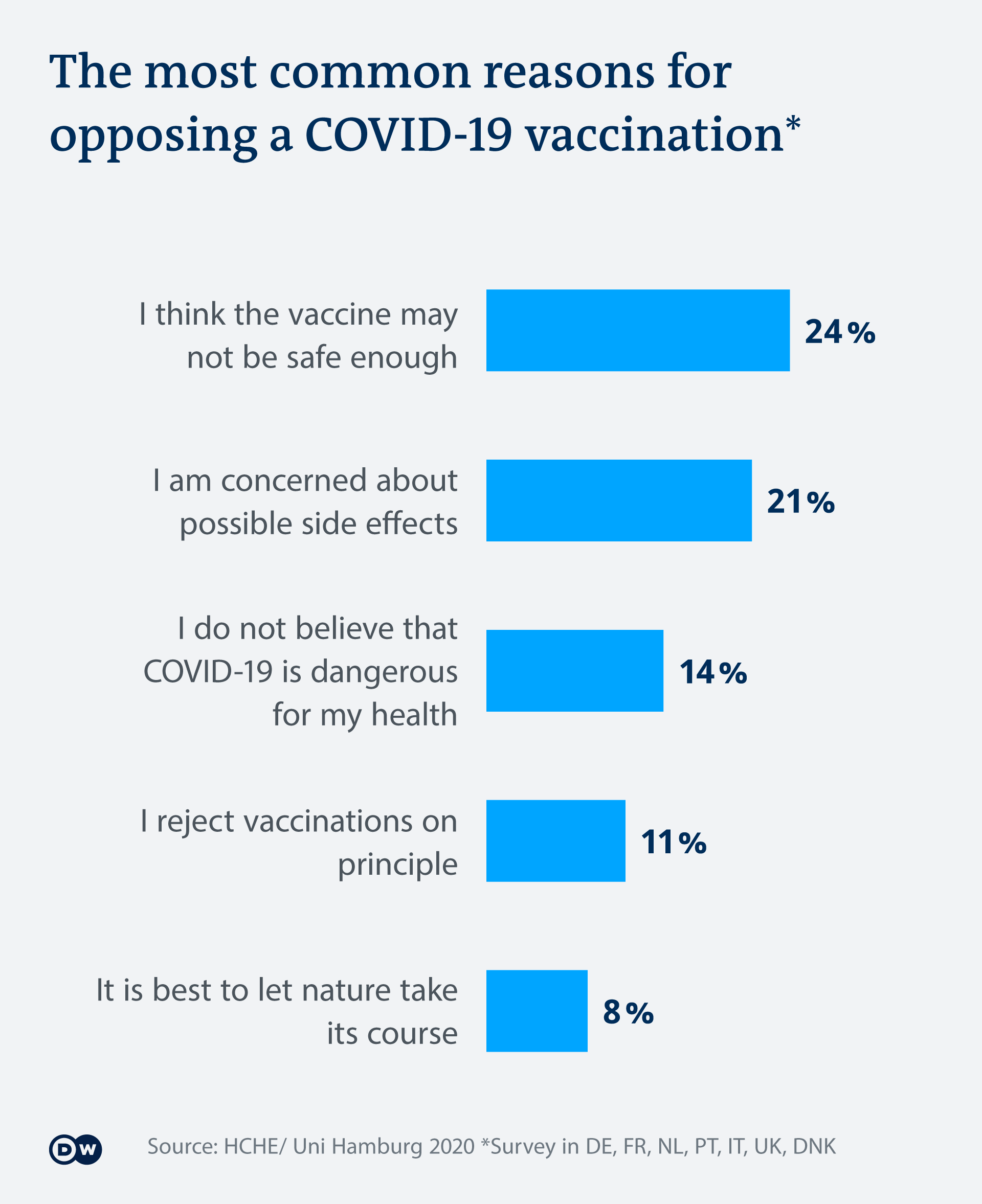 Covid 19 Difficult Trade Offs In Astrazeneca Vaccine Development Science In Depth Reporting On Science And Technology Dw 28 10 2020
Covid 19 Difficult Trade Offs In Astrazeneca Vaccine Development Science In Depth Reporting On Science And Technology Dw 28 10 2020
 A Phase Iii Study Of Triple Therapy With Budesonide Glycopyrrolate Formoterol Fumarate Metered Dose Inhaler 320 18 9 6 Mg And 160 18 9 6 Mg Using Co Suspension Delivery Technology In Moderate To Very Severe Copd The Ethos Study Protocol Respiratory
A Phase Iii Study Of Triple Therapy With Budesonide Glycopyrrolate Formoterol Fumarate Metered Dose Inhaler 320 18 9 6 Mg And 160 18 9 6 Mg Using Co Suspension Delivery Technology In Moderate To Very Severe Copd The Ethos Study Protocol Respiratory
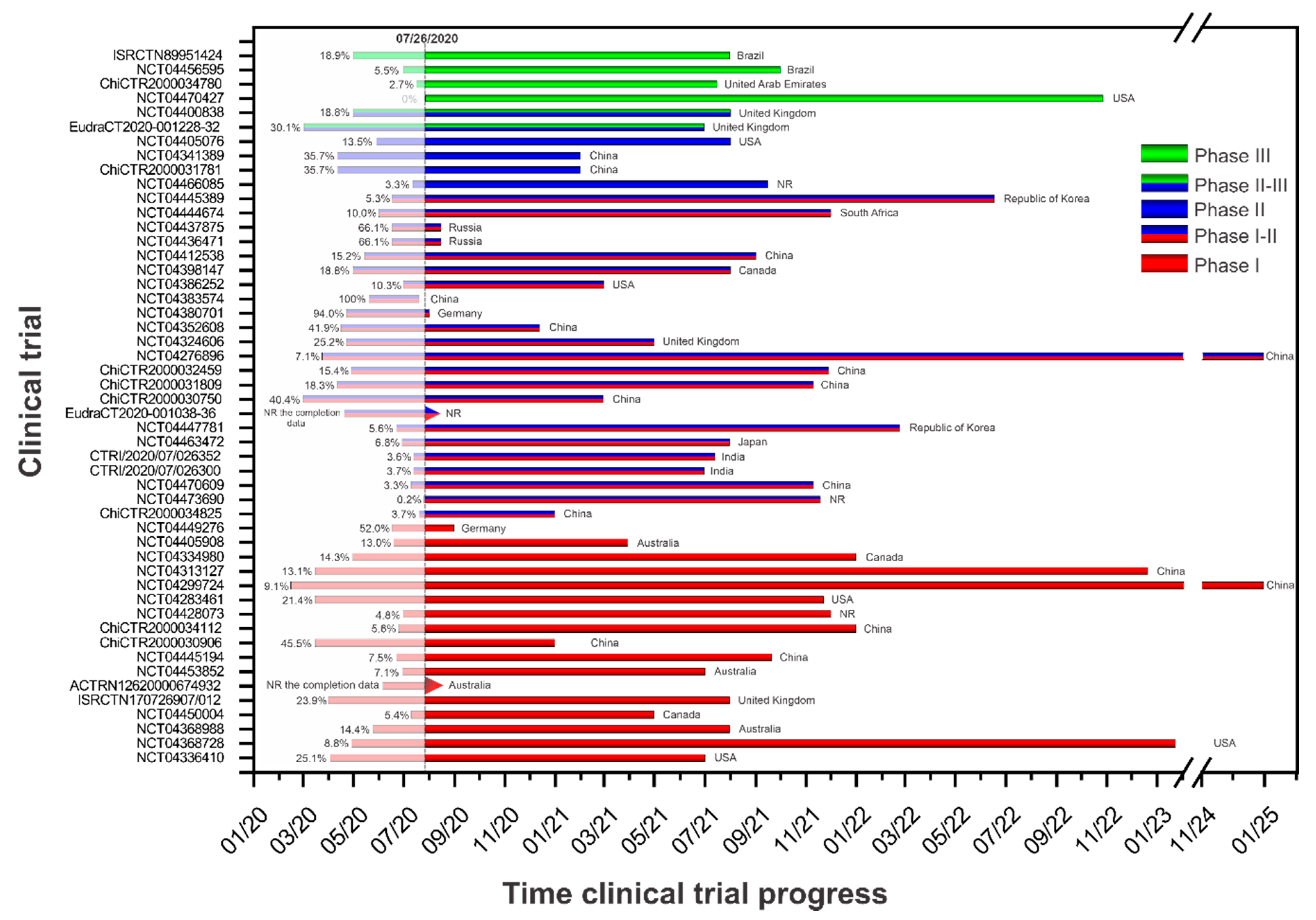 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
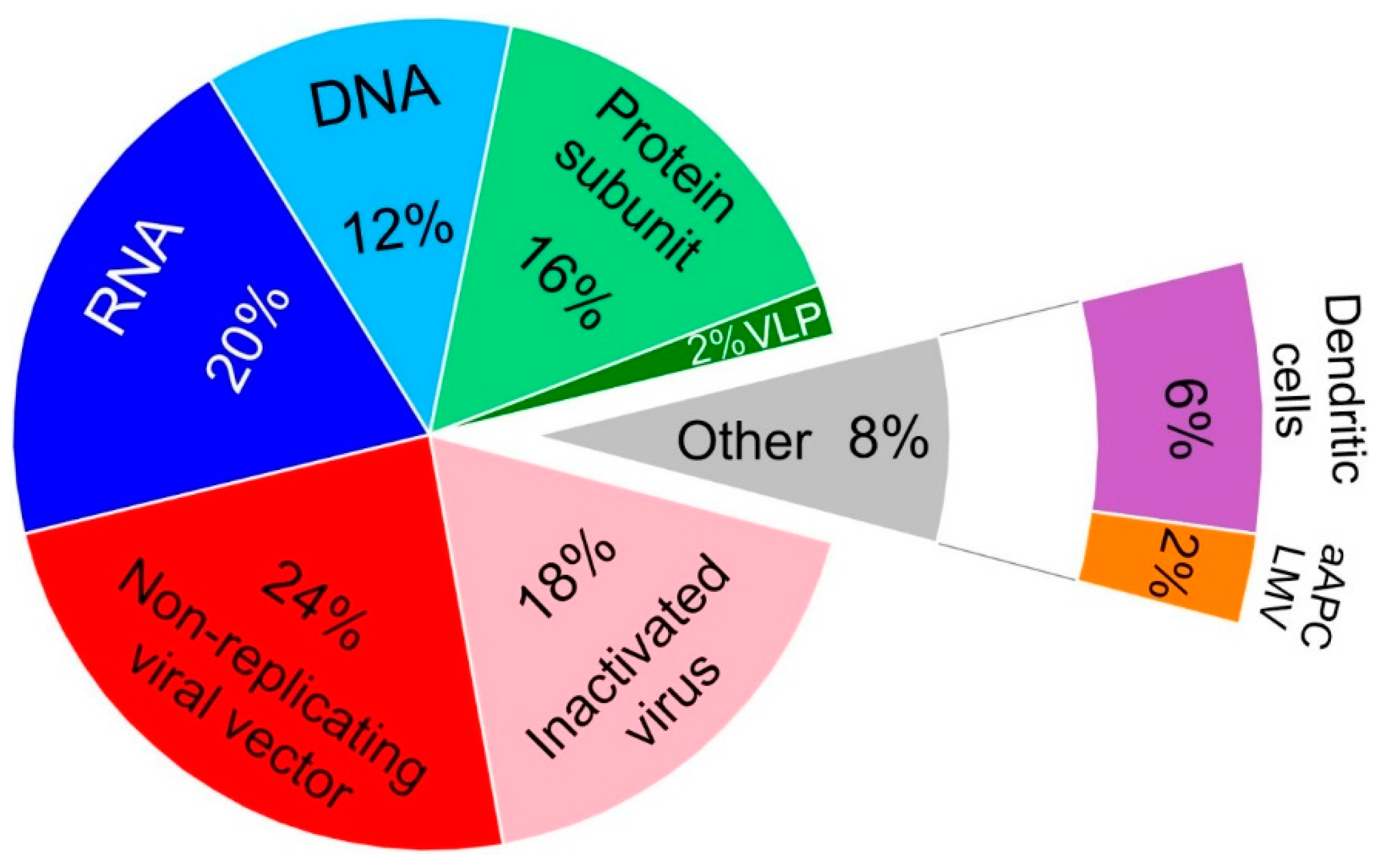 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
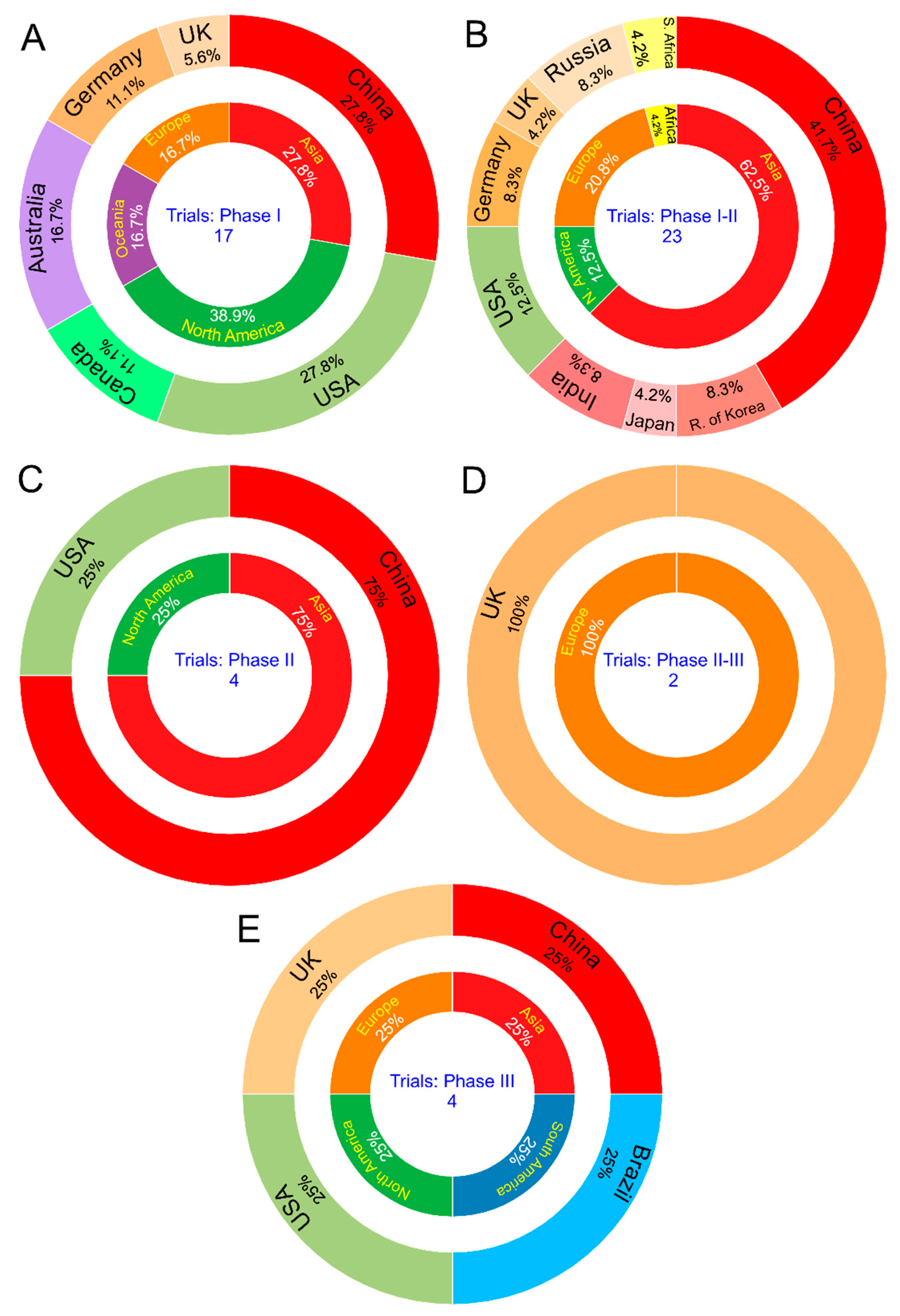 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
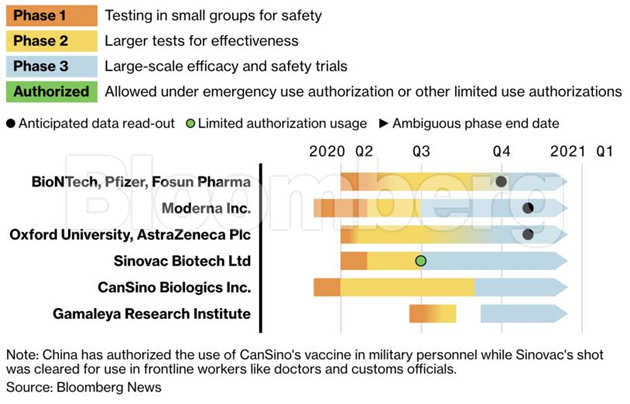
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
Post a Comment for "Astrazeneca Phase 3 Results Date"