Astrazeneca Vaccine Phase 3 End Date
The interim analysis published today in. Actual Study Start Date.
 Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
Pfizer And Biontech S Covid 19 Vaccine Found To Be 90 Effective Financial Times
Participants aged 18 years and older were randomly assigned 11 to ChAdOx1 nCoV-19 vaccine or control meningococcal group A C W and Y conjugate vaccine or saline.

Astrazeneca vaccine phase 3 end date. 20072020 The companies plan to seek regulatory review as early as October 2020 manufacture globally up to 100 million doses by the end of 2020 and more than 13 billion doses by the end. While AstraZeneca is the sponsor of late-phase AZD1222 trials in the US. Until the end of October 2020 the AstraZeneca vaccine was able to reduce a persons risk of COVID-19 becoming infected with the virus just 14 days after injection but.
Estimated Study Completion Date. Actual Primary Completion Date. Approximately 42 of global participants and 30 of US.
Trial found its vaccine 79 effective but faced backlash after NIAID said Tuesday it may have included information that provided an incomplete view of the efficacy data. A Phase III Open-label Study in Adults to Determine the Safety and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. This remarkable achievement is much-needed good news as COVID-19 cases are currently at their highest daily.
25032021 AstraZeneca on Thursday published an updated analysis confirming that a Phase 3 trial involving its COVID-19 vaccine was safe and effective and showed to. 2020 has been a difficult year for all but has seen 58 vaccines against severe acute respiratory syndrome coronavirus 2 SARS-CoV-2 be developed and in clinical trials1 with some vaccines reportedly having more than 90 efficacy against COVID-19 in clinical trials. 20042021 AstraZeneca reports good progress of Covid-19 vaccine in trials 31 Jul 2020 Last Updated July 31st 2020 0715 AstraZeneca has reported that it has obtained good data so far on its Covid-19 vaccine candidate which was licensed from the University of Oxford and is currently in large-scale clinical studies.
Estimated Primary Completion Date. Estimated Study Completion Date. Participants have racially and ethnically diverse backgrounds and 41 of global and 45 of US.
How well the vaccine works in the elderly and other vulnerable groups. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. 08032021 Data are expected soon in an early February interview AstraZeneca research chief Mene Pangalos had predicted results were four to six weeks away and could provide crucial answers to the remaining questions.
This analysis includes data from four ongoing blinded randomised controlled trials done across the UK Brazil and South Africa. Furthermore the vaccine is close to being finalised for approval US. Heres what to watch.
24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US. On 23 November 2020 the first interim data was released by Oxford University and AstraZeneca from the vaccines ongoing Phase III trials. 25032021 AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety.
The interim data reported a 70 efficacy based on combined results of 62 and 90 from different groups of. The Phase 3 clinical trial of BNT162b2 began on July 27 and has enrolled 43661 participants to date 41135 of whom have received a second dose of the vaccine candidate as of November 13 2020. With a 76 efficacy rate against symptomatic COVIDFlashback.
04092020 Coronavirus vaccine by AstraZeneca has reached phase 3 clinical trials in the US. Participants are 56-85 years. And Russia Oxford the originator of the vaccine is taking the lead on phase 3 studies in Brazil and the UK.
Actual Study Start Date. The company reported on Monday that a US. 19112020 Phase 3 trials of the Oxford-AstraZeneca COVID-19 vaccine are now accumulating the data needed to report results as a renewed surge of the pandemic hits countries around the world says Oxford.
The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
 The Astrazeneca Phase 3 Covid 19 Vaccine Study Unprecedented Compression Of Timeline Of Est Start Date To Est Primary Completion Date
The Astrazeneca Phase 3 Covid 19 Vaccine Study Unprecedented Compression Of Timeline Of Est Start Date To Est Primary Completion Date
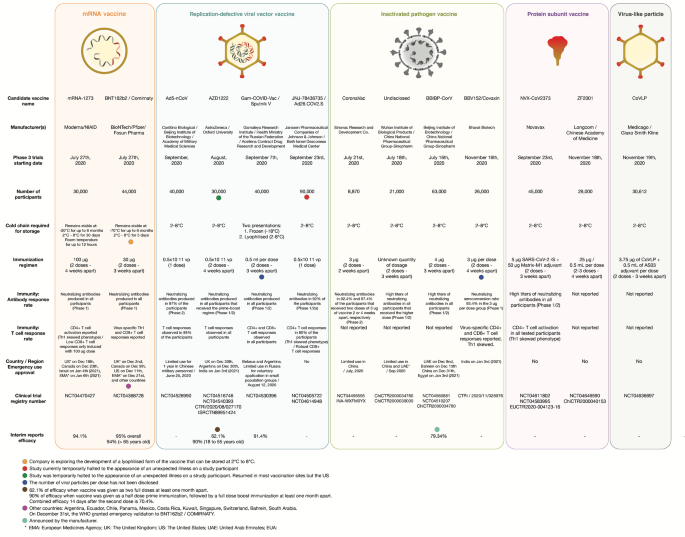 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek

 Infocus Unanswered Questions On The Covid 19 Vaccine Efg Asset Management
Infocus Unanswered Questions On The Covid 19 Vaccine Efg Asset Management
 Covid 19 Vaccines November Update Progress Of Clinical Trials Post
Covid 19 Vaccines November Update Progress Of Clinical Trials Post
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
 Hopes Rise For End Of Pandemic As Pfizer Says Vaccine Has 90 Efficacy Coronavirus The Guardian
Hopes Rise For End Of Pandemic As Pfizer Says Vaccine Has 90 Efficacy Coronavirus The Guardian
 Russian Covid 19 Vaccine Sputnik V Under Phase 3 Trials In India To Cost Less Than 10 Per Dose News
Russian Covid 19 Vaccine Sputnik V Under Phase 3 Trials In India To Cost Less Than 10 Per Dose News
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post
Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Covid 19 Race For A Vaccine Deutsche Bank
Covid 19 Race For A Vaccine Deutsche Bank
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Astrazeneca Reports Up To Date Covid 19 Vaccine Efficacy Results
Post a Comment for "Astrazeneca Vaccine Phase 3 End Date"