Astrazeneca Phase 3 Vaccine Covid
In the UK the rollout of the Pfizer-BioNTech and Oxford-AstraZeneca COVID-19 vaccines began on the 8 th of December 2020 and the 4 th of. The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
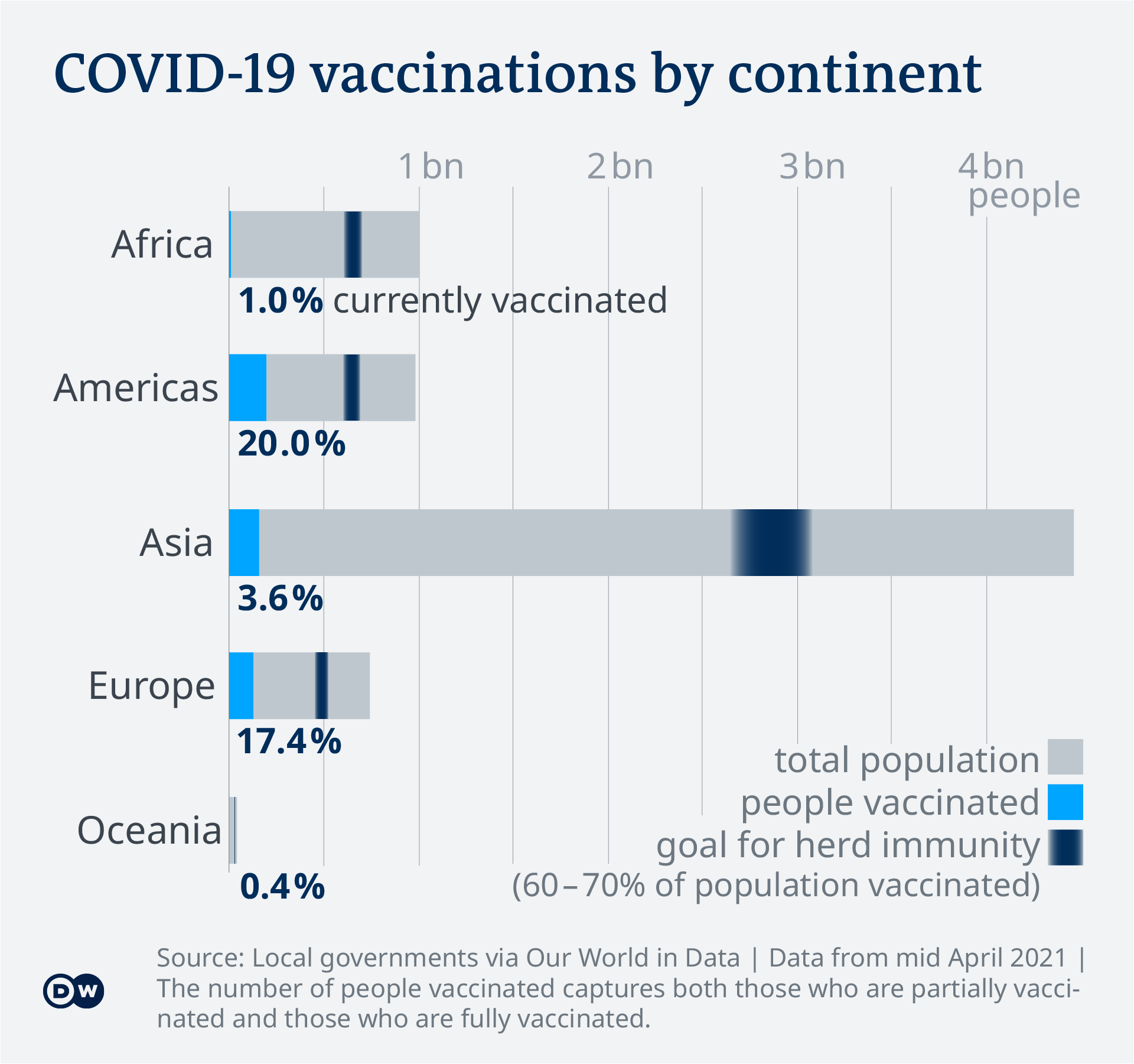 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
22032021 Results of the trial which involved more than 32000 volunteers showed two doses of the vaccine administered four weeks apart had an efficacy of 79 at preventing symptoms of.

Astrazeneca phase 3 vaccine covid. Un vaccino anti COVID-19 sperimentale a vettore basato sulladenovirus ChAdOx1 sviluppato presso il Jenner Institute dellUniversit. 1 giorno fa So far the COVID-19 vaccines are procured by the government and administered for free to the public. Actual Study Start Date.
Developed by Oxford University and AstraZeneca it is given by intramuscular injection using as a vector the modified chimpanzee adenovirus ChAdOx1. The regulators have already approved the Pfizer-BioNTech vaccine Chinas Sinovac and Britains AstraZeneca. Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704.
So far we have only seen around 24 of the population that has obtained at least one dose of the vaccine. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing accuracy questions earlier this week surrounding a preliminary report from its US. 28072020 The government has chosen five sites to initiate the third and final phase of much-awaited Oxford-AstraZeneca COVID-19 vaccine candidate before itll finally be administered to Indians.
15 ore fa UK government decided to delay second vaccine doses. What we know about blood clots side effects and risks for under-30s The UK medicines regulator says the benefits of the AstraZeneca vaccine continue to outweigh any. AstraZeneca committed to not profit from the Covid-19 vaccine during the pandemic.
31082020 CNN British drugmaker AstraZeneca said Monday it has started Phase 3 trials of its experimental coronavirus vaccine in the United States. 20042021 Serum Institute of India filed an application with the Drugs Controller General of India DCGI to study the Covid-19 vaccine candidate in Phase II and III trials. Il vaccino anti COVID-19 AstraZeneca o vaccino anti COVID-19 Oxford-AstraZeneca nome in codice AZD1222 nome commerciale Vaxzevria.
UPDATED ON OCT 24 2020 1246 PM IST The Brihanmumbai Municipal Corporation BMC has completed the second phase of clinical trials of the Oxford-AstraZeneca Covid-19 vaccine. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19.
1 giorno fa THERE is no evidence that women are at greater risk from blood clots after the AstraZeneca coronavirus vaccine MPs have been toldBut as extremely ra. Di Oxford in collaborazione con la societ. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19.
19012021 For these reasons even when COVID-19 vaccines have achieved licensure via current phase 3 trials there will be substantial uncertainties about how useful the vaccines will. Estimated Study Completion Date. AstraZeneca e litaliana IRBM Science Park.
The efficacy of the vaccine is 760 at preventing symptomatic COVID-19 beginning at 22 days. 1 giorno fa COVID-19 vaccine phase 3. Actual Primary Completion Date.
11 ore fa Astrazeneca Covid vaccine QA. Anxious Indians are complaining of CoWIN and Aarogya Setu registration being delayed Zomato reveals its much-awaited IPO. 1 giorno fa Data from the ZOE Covid Symptom Tracking Study showed fewer people experienced mild side effects from AstraZeneca and Pfizers vaccines than had been suggested in clinical trials.
.jpg) Uk Public Health Authorities Review Early Data On The Effectiveness Of Pfizer And Astrazeneca Covid 19 Vaccines
Uk Public Health Authorities Review Early Data On The Effectiveness Of Pfizer And Astrazeneca Covid 19 Vaccines
 Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
Vaccino Astrazeneca 100 Efficacia Nel Prevenire Casi Gravi E No Aumento Rischio Trombosi Lo Studio Usa
 Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
 Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India
Covid 19 Vaccine Latest Updates Astrazeneca S Manufacturing Error Raises Concerns On Vaccine Efficacy Covaxin To Begin Phase Iii Trials In Gujarat Business Insider India
 Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
 Covid 19 Vaccine These Countries Have Started Rolling Out Coronavirus Vaccine Check Full List Here
Covid 19 Vaccine These Countries Have Started Rolling Out Coronavirus Vaccine Check Full List Here
 Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post
Astrazeneca Ceo Says Coronavirus Vaccine Could Be Ready In December South China Morning Post

 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
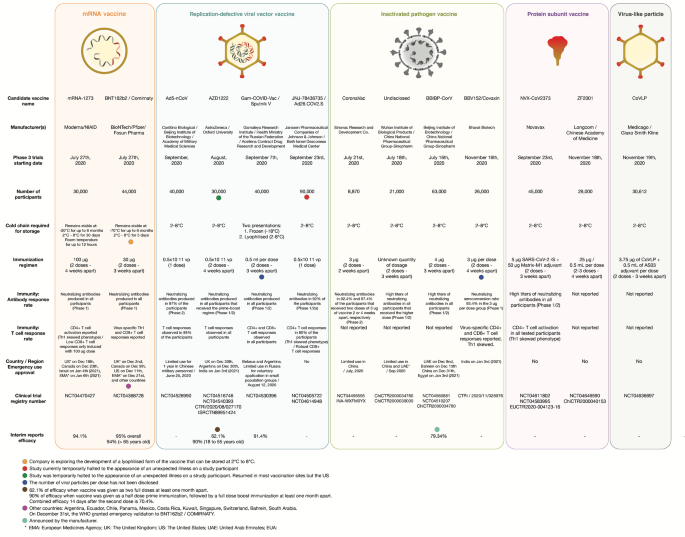 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines

 Covid 19 Vaccine Trial Will Continue After Volunteer Death Science Business
Covid 19 Vaccine Trial Will Continue After Volunteer Death Science Business
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
Covid 19 Italy Bans Batch Of Astrazeneca Vaccine The Local
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
Post a Comment for "Astrazeneca Phase 3 Vaccine Covid"