Astrazeneca Vaccine Phase 3 Results
The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19. AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US.
 Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
Coronavirus Vaccine Update New Phase 2 Trial Results Of Oxford Covid 19 Vaccine Show Success In Older People
Actual Primary Completion Date.
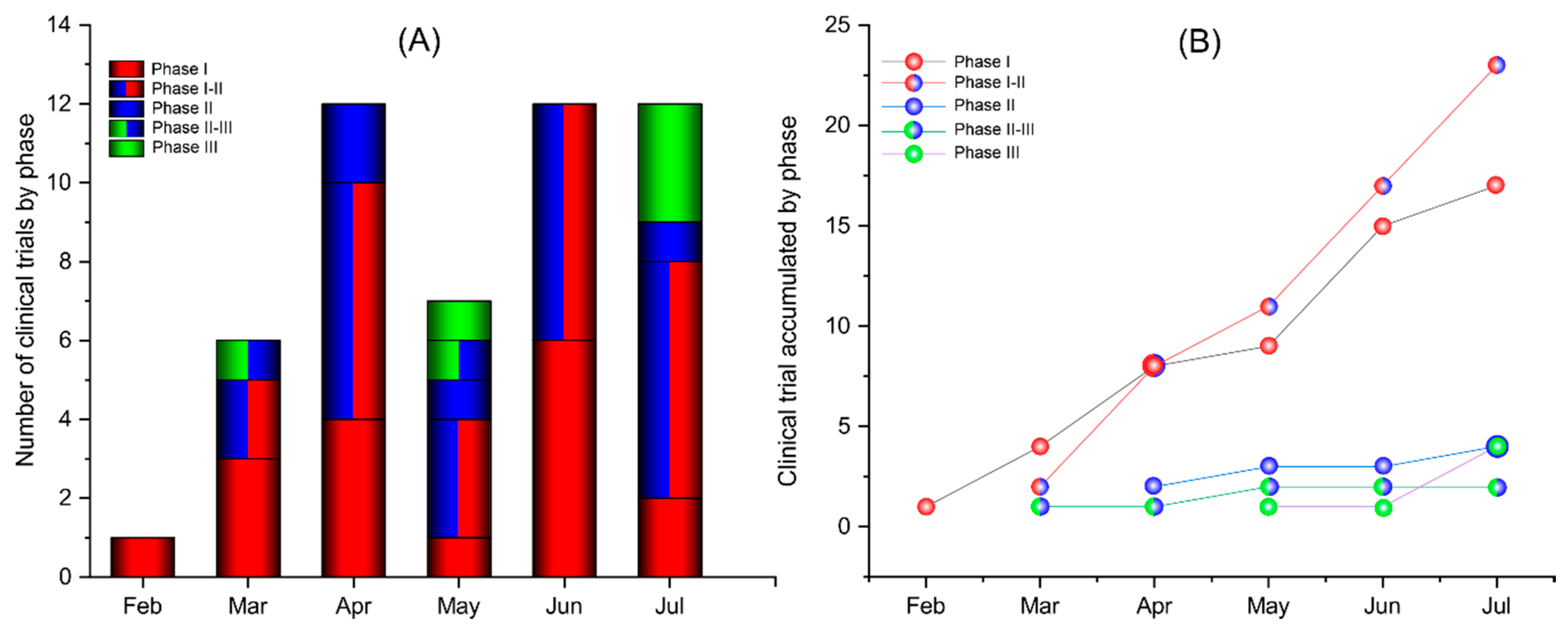
Astrazeneca vaccine phase 3 results. 22032021 AstraZeneca today announced interim results of their phase 3 US study which indicate 79 overall efficacy of their vaccine against symptomatic COVID-19 80 in people aged 65 years and older and. Findings were published in The Lancet journal. AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US.
Modernas phase 3 analysis involved 30000 people in the US half given the vaccine and. SeventyFour iStock The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. Rather than results from one large phase 3 trial which is what we saw from trials of other covid-19 vaccines these results were actually pooled data from two separate trials one in the UK and.
Another vaccine option in coming months. Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation. The efficacy of the vaccine is 760 at preventing symptomatic COVID-19 beginning at 22 days following the first dose and 813 after the second dose.
20042021 In the Phase III COV001 trial conducted in the UK the vaccine was tolerated and generated strong immune responses in all evaluated participants. Oxford and AstraZeneca researchers present a pooled analysis in The Lancet of Phase 3 trials of a coronavirus vaccine resulting in an average efficacy of 704. A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19.
Estimated Study Completion Date. The vaccine has a good safety profile with side effects including injection-site pain headache and. Tuesdays results the first Phase 3 clinical data for a.
The interim analysis for efficacy was based on 11636 participants accruing 131. 30112020 The UK has bought 100m doses of the Oxford vaccine which will be a core part of its mass immunisation plans. Another vaccine option in coming months.
First peer-reviewed results of phase 3 human trials of Oxford coronavirus vaccine demonstrate efficacy University of Oxford. 20072020 According to AstraZeneca late-stage trials of AZD1222 include a Phase III trial in Brazil 2000 participants ISRCTN89951424 a Phase IIbIII trial in. Just last week British scientists published a paper showing a single dose lowered the rate of symptomatic illness in the elderly in the UK.
04022021 AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing Covid-19 more than 22 days after the first dose. Study results showed that a single dose of the vaccine led to a four-fold increase in antibodies to the SARS-CoV-2 spike protein in 95 of participants one month following. But in order to do so the company needs to.
Actual Study Start Date. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. 08032021 AstraZeneca has pointed to early-stage results showing immune responses in older and younger volunteers are comparable with fewer side effects in the elderly.
08122020 Topline data on the AstraZeneca vaccine from a Phase 3 trial were released in November. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa.
10032021 Vaccine major Bharat Biotech submitted the initial results of the phase 3 trials of Covaxin its anti-Coronavirus disease Covid-19 vaccine that has shown nearly 81 efficacy against the viral. New vaccine efficacy results are reported now in The Lancet. But in order to do so the company needs.
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
 Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
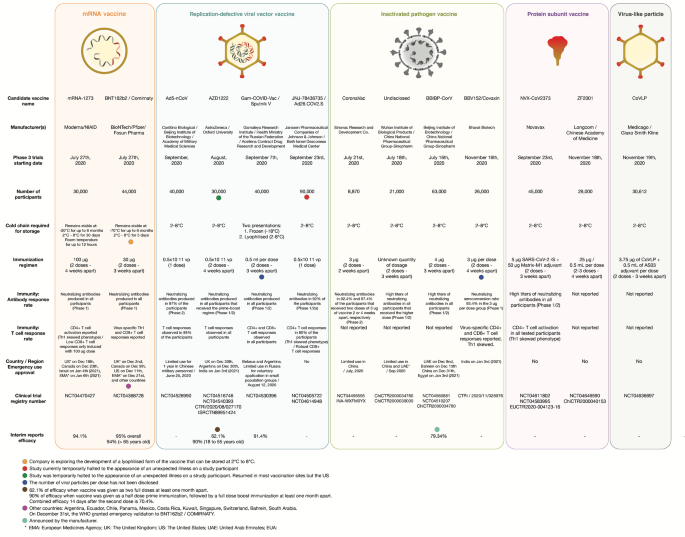 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
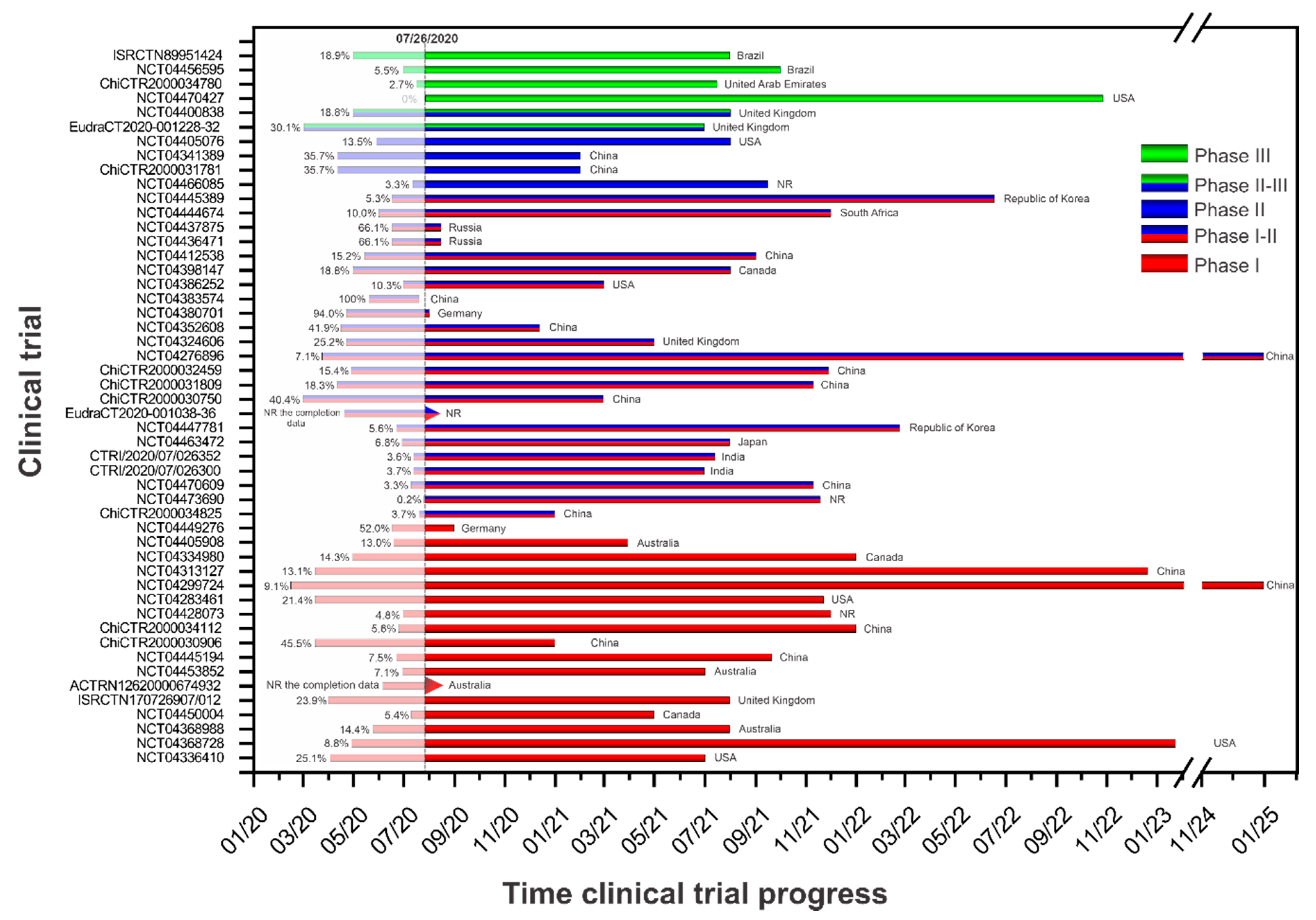 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
Update Astrazenca Oxford Vaccine 79 Effective In Us Phase 3 Interim Analysis
 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
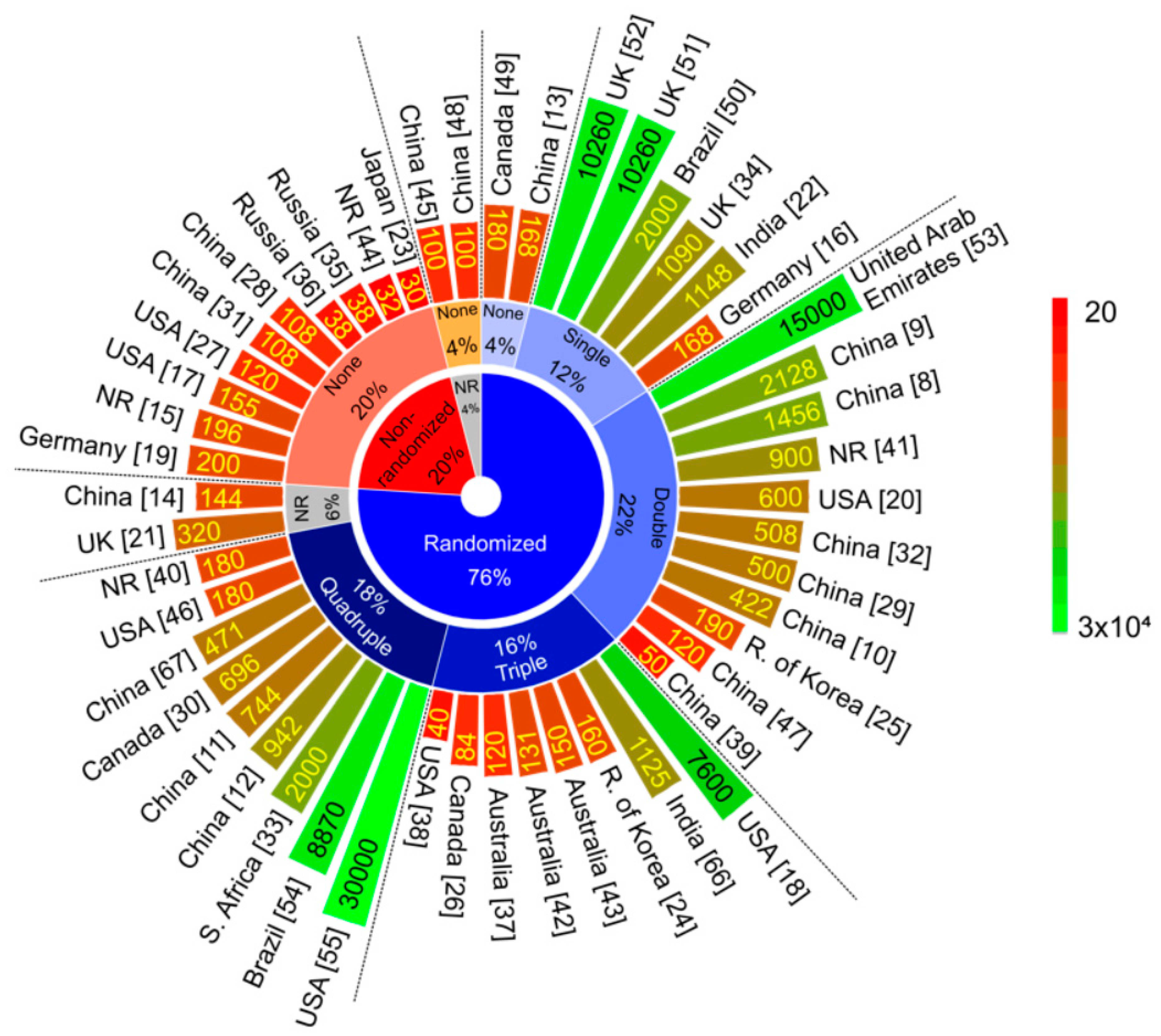 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Sinopharm S Covid 19 Vaccine Scores Approval In China Pmlive
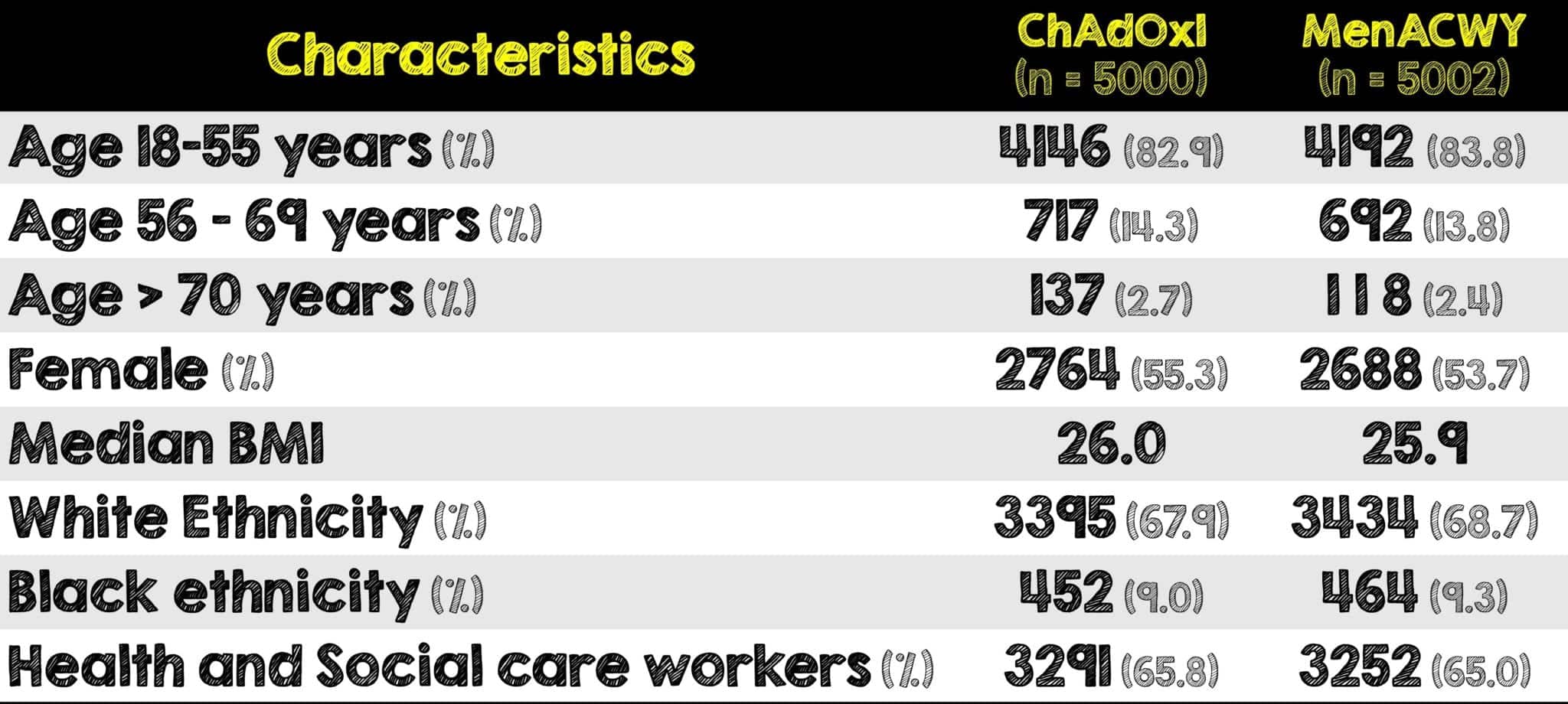 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
Coronavirus Vaccine Update What Is Oxford Astrazeneca Covid 19 Vaccine Error All You Need To Know The Financial Express
 Sputnik V On Twitter 1 7 Currently There Are Only 4 Vaccines In The World That Published Phase 3 Results In This Order Pfizer Sputnik V Moderna Astrazeneca Sputnikv Has Significant Advantages In
Sputnik V On Twitter 1 7 Currently There Are Only 4 Vaccines In The World That Published Phase 3 Results In This Order Pfizer Sputnik V Moderna Astrazeneca Sputnikv Has Significant Advantages In
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Astrazeneca To Give 400m Covid 19 Vaccine Doses To Europe Data Expected August September 2020 06 14 Bioworld
Astrazeneca To Give 400m Covid 19 Vaccine Doses To Europe Data Expected August September 2020 06 14 Bioworld
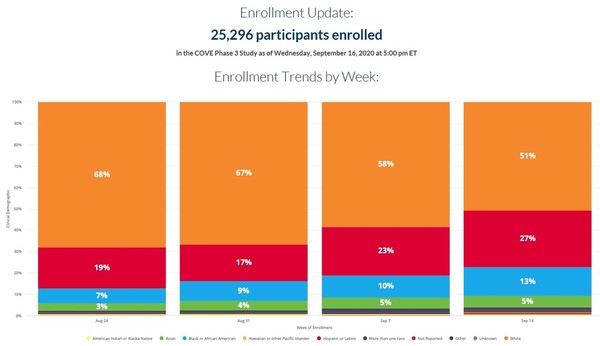
Post a Comment for "Astrazeneca Vaccine Phase 3 Results"