Astrazeneca Phase 3 Vaccine Trial
04022021 AstraZenecas Covid-19 vaccine proves safe and effective in Phase III trial. 31082020 Currently Phase 3 trials of AstraZenecas vaccine are going on in Britain Brazil and South Africa.
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
The trial funded by the.

Astrazeneca phase 3 vaccine trial. Has been put on hold due to a suspected serious. Phase 3 clinical trial of its COVID-19 vaccine candidate AZD1222. 31 it has begun enrolling up to 30000 people in a phase 3 trial for its experimental COVID-19 vaccine.
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. Trial is currently taking place at 62 sites across the country according to clinicaltrialsgov a. Actual Primary Completion Date.
14102020 CNN AstraZeneca whose Phase 3 coronavirus vaccine clinical trial has been on hold for more than a month did not get critical safety data to. 19012021 An important secondary objective of phase 3 randomized trials will be testing of immune serum from volunteers who were vaccinated and became infected compared to a matched set of vaccinated. 24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing criticism earlier this week over a preliminary report from its US.
Actual Study Start Date. Added value of this study. The company now says.
AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing Covid-19 more than 22 days after the first dose. The Biomedical Advanced Research and Development Authority-funded study will enroll 30000 adults at. AstraZeneca has begun a US.
We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa. 25032021 AstraZeneca announced today its results from the phase 3 clinical trial that it carried out in the US Chile and Peru. Trials are also planned for Japan and Russia.
08092020 A large Phase 3 study testing a Covid-19 vaccine being developed by AstraZeneca and the University of Oxford at dozens of sites across the US. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun.
Researchers at the University of Oxford have begun enrolling subjects in a phase 23 clinical trial of AstraZeneca-partnered COVID-19 vaccine AZD1222. Estimated Study Completion Date. 09092020 AstraZeneca only began its Phase 3 trial in the US.
22032021 Astrazeneca vaccine in phase three trial March 22 2021 645 PM AstraZeneca is in phase three trial with its vaccine and looks to file for emergency use with the FDA next month. This is a new trial required for approval of the vaccine for use in the US in consequence of the companys amateurish mismanagement of its original clinical trial. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704.
29102020 The vaccine developed by Oxford University and AstraZeneca is called AZD1222 and has successfully gone through earlier smaller phase. New vaccine efficacy results are reported now in The Lancet. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 COVID-19.
The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use.
 Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
 Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
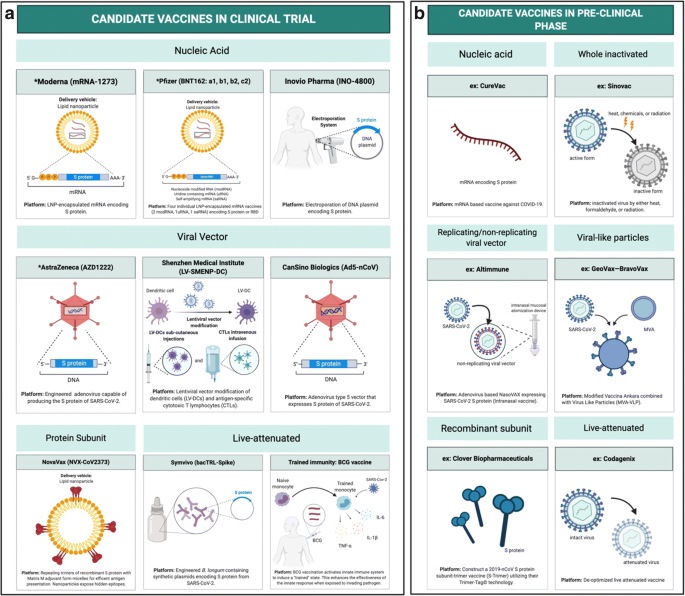 The Impact Of Immuno Aging On Sars Cov 2 Vaccine Development Springerlink
The Impact Of Immuno Aging On Sars Cov 2 Vaccine Development Springerlink
 Astrazeneca S Covid 19 Vaccine Candidate Begins Phase 3 Trial Rt
Astrazeneca S Covid 19 Vaccine Candidate Begins Phase 3 Trial Rt
 Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3

 Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
Phase 3 Trials Begin For Novavax Vaccine In Us Mexico Industryweek
 Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
 Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
 Oxford Astrazeneca Covid Vaccine Prompts Immune Response In All Adults
Oxford Astrazeneca Covid Vaccine Prompts Immune Response In All Adults
 Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 Covid 19 Vaccine Trial Will Continue After Volunteer Death Science Business
Covid 19 Vaccine Trial Will Continue After Volunteer Death Science Business
 Coronavirus Covid 19 Vaccine Latest Update September 2 Oxford Begins Stage 3 Trials In The Us
Coronavirus Covid 19 Vaccine Latest Update September 2 Oxford Begins Stage 3 Trials In The Us
 Astrazeneca Covid 19 Vaccine Completes Phase 3 Trial Showing 79 Percent Effectiveness
Astrazeneca Covid 19 Vaccine Completes Phase 3 Trial Showing 79 Percent Effectiveness
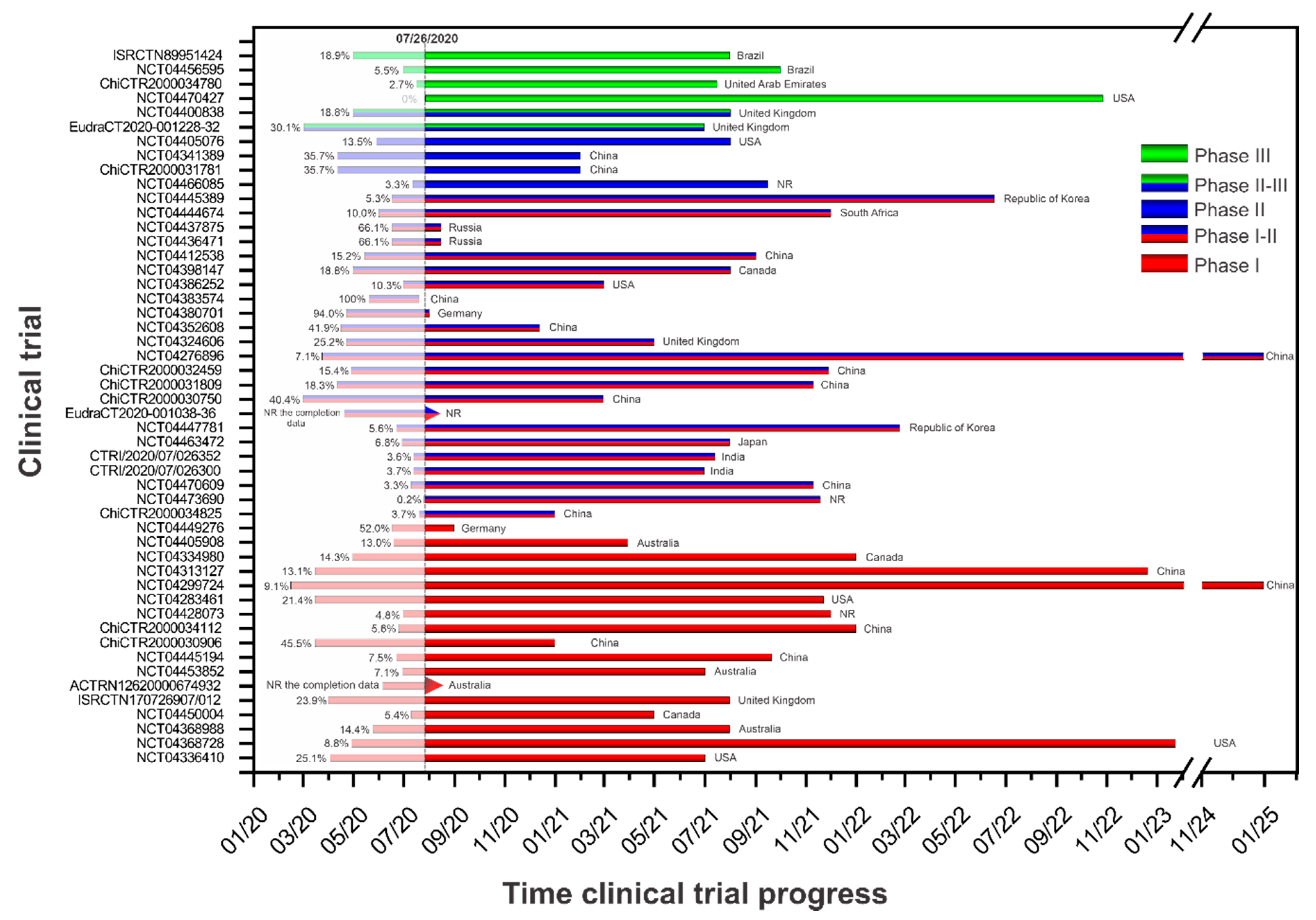 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Post a Comment for "Astrazeneca Phase 3 Vaccine Trial"