Astrazeneca Phase 3 Results Uk
It was a 15000-participant trial in the United Arab Emirates UAE of the 2 Sinopharm inactivated virus vaccines an old school type of vaccine. 20072020 The results published in The Lancet confirmed a single dose of AZD1222 resulted in a four-fold increase in antibodies to the SARS-CoV-2 virus spike protein in 95 of participants one month after injection.
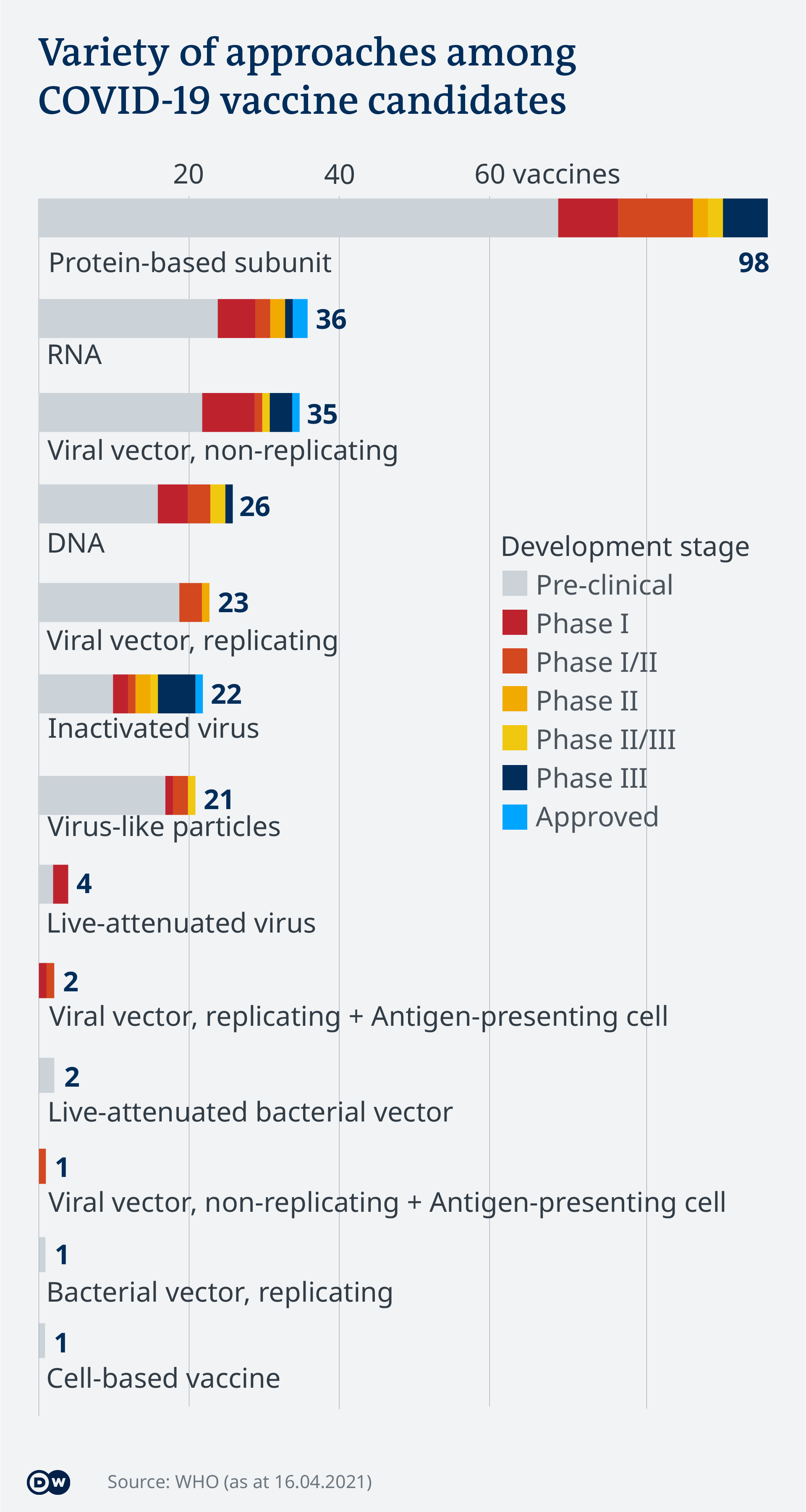 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
23112020 COV002 is a single-blinded multi-centre randomised controlled Phase IIIII trial assessing the safety efficacy and immunogenicity of AZD1222 in 12390 participants in the UK.

Astrazeneca phase 3 results uk. Just last week British scientists published a paper showing a single dose lowered the rate of symptomatic illness in the elderly in the UK. Trial participants to date are aged 18 years or over who are healthy or have medically stable chronic diseases and are at increased risk for being exposed to the SARS-CoV-2 virus. New vaccine efficacy results are reported now in The Lancet.
Results of an interim analysis of the Phase III programme conducted by Oxford University with AZD1222 peer-reviewed and published in The Lancet today demonstrated that the vaccine is safe and effective at preventing symptomatic COVID-19 and that it protects against severe disease and hospitalisation. And Brazil and in a Phase 12 trial in South Africa. 3-min read AstraZeneca AZN anticipates having Phase 3 trial results by the end of March potentially giving the US.
The first full peer-reviewed results of phase 3 trials of the COVID-19 vaccine developed by AstraZeneca and Oxford University show that it is safe and up to 90 effective in preventing infection supporting regulatory submissions for emergency use. 25032021 AstraZeneca issues new Phase 3 data for COVID vaccine confirming safety and efficacy. A large United Kingdom-based Phase 23 study testing a Covid-19 vaccine being developed by AstraZeneca has been restarted according to a statement from the company.
11092020 The first fully-recruited phase 3 trial plus likely first results on Covid-19 protection. The new study published in the Lancet is the first peer-reviewed publication of phase 3 data from studies of a vaccine against the coronavirus. 11 February 2021 1204 pm.
08032021 AstraZeneca has pointed to early-stage results showing immune responses in older and younger volunteers are comparable with fewer side effects in the elderly. Another vaccine option in. The Lancet has published the worlds first peer-reviewed results of the University of Oxford and AstraZeneca phase 3 COVID-19 vaccine trials.
We report on the first clinical efficacy results of ChAdOx1 nCoV-19 in a pooled analysis of phase 23 trials in the UK and Brazil and safety data from more than 20 000 participants enrolled across four clinical trials in the UK Brazil and South Africa. Results demonstrated vaccine efficacy of 76 CI. The interim analysis for efficacy was based on.
AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety. Based on the RML data a Phase 1 trial of the candidate vaccine began on April 23 in healthy volunteers in the UK. 3 inactivated virus vaccines The first phase 3 trial to reach full enrolment was in the middle of August.
Investigators recently reported promising results in The Lancet. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. 22032021 The AstraZeneca US Phase III trial of AZD1222 demonstrated statistically significant vaccine efficacy of 79 at preventing symptomatic COVID-19 and 100 efficacy at preventing severe disease and hospitalisation.
In all participants a T-cell response was induced peaking by day 14 and maintained two months after injection. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature. The vaccine has been supported by UK Research and Innovations Medical Research Council MRC and the published data confirms the vaccine is safe effective and gives good protection.
This interim safety and efficacy analysis was based on 32449 participants accruing 141 symptomatic cases of COVID-19. Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704. Rather than results from one large phase 3 trial which is what we saw from trials of other covid-19 vaccines these results were actually pooled data from two separate trials one in the UK and.
With a 76 efficacy rate against symptomatic COVID. Doses Two 28 days apart. Currently the vaccine candidate is being evaluated in Phase 23 trials in the UK.
Efficacy Phase 3 trials suggest 893. 07042021 Efficacy Phase 3 trial results suggest 941. Janssen part of Johnson.
03022021 The primary analysis of the Phase III clinical trials from the UK Brazil and South Africa published as a preprint in The Lancet confirmed COVID-19 Vaccine AstraZeneca is safe and effective at preventing COVID-19 with no severe cases and no hospitalisations more than 22 days after the first dose. Efficacy results are based on data taken from 11636 volunteers across the United Kingdom and Brazil.
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
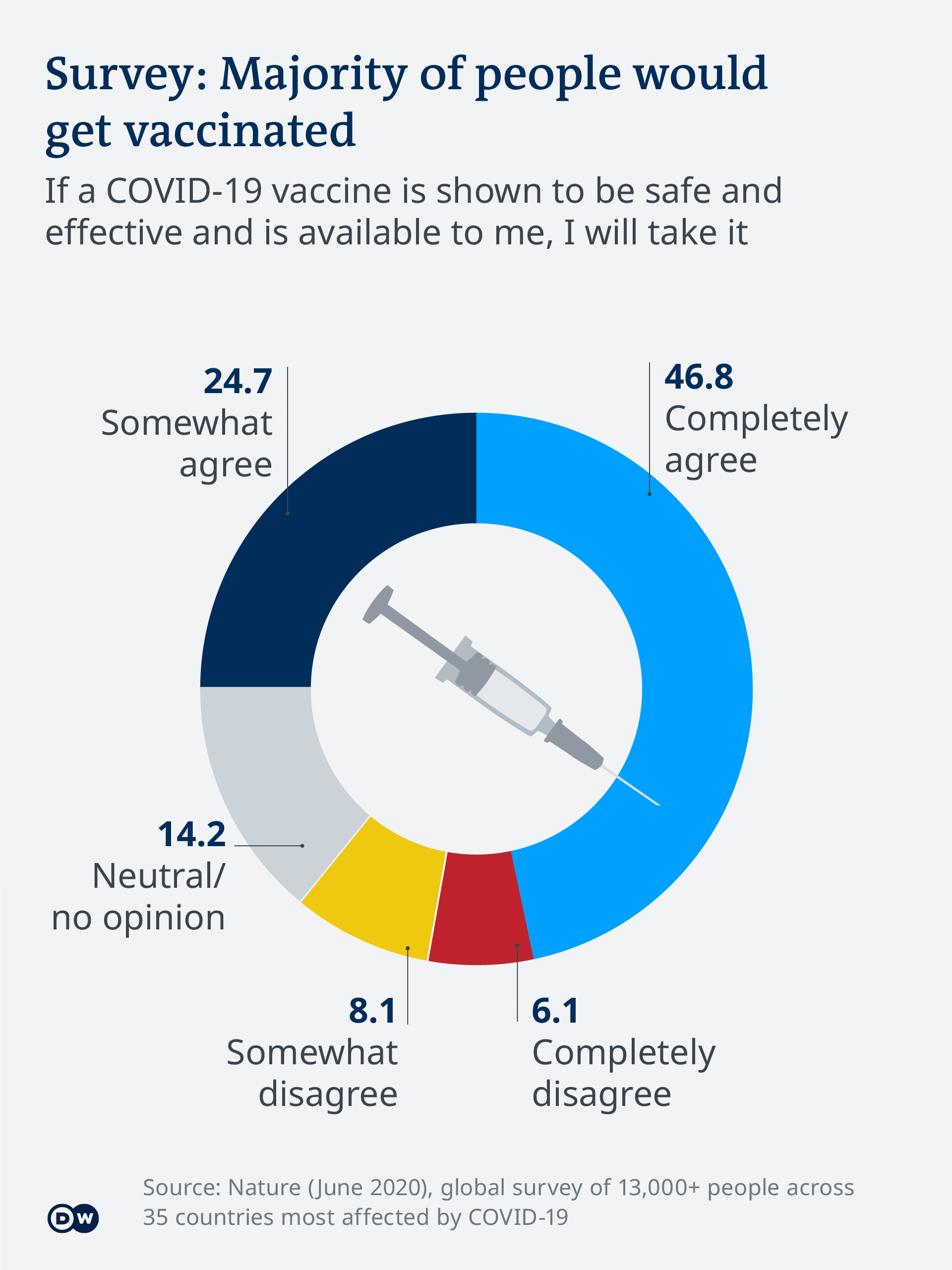 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Covid Vaccine Astrazeneca Phase Iii Trials Interim Results Youtube
Covid Vaccine Astrazeneca Phase Iii Trials Interim Results Youtube
 Covid 19 Vaccines Vaccine Knowledge
Covid 19 Vaccines Vaccine Knowledge
 Astrazeneca Merck Co S Lynparza Gets Eu Approval In Prostate Cancer S P Global Market Intelligence
Astrazeneca Merck Co S Lynparza Gets Eu Approval In Prostate Cancer S P Global Market Intelligence
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
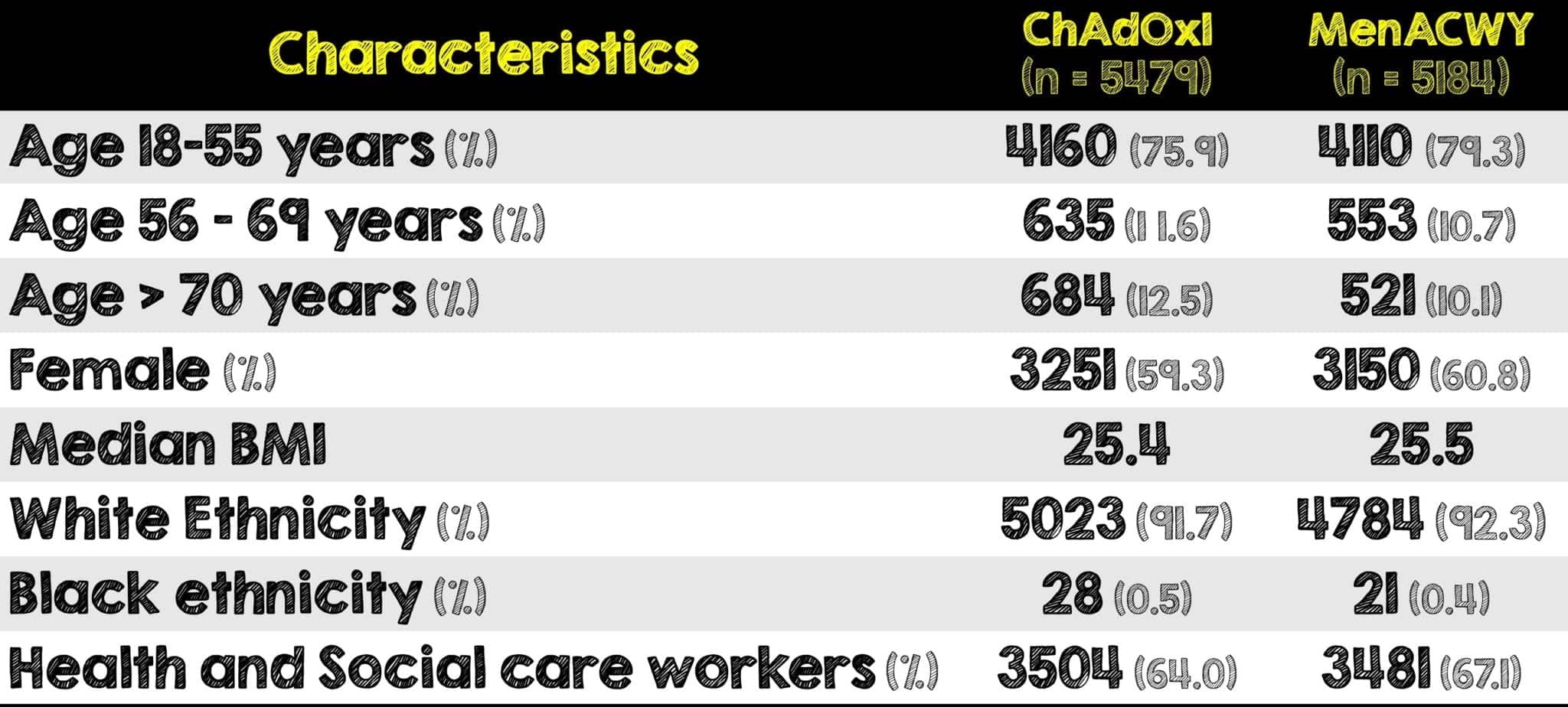 Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
Covid 19 Update The Chadox1 Astrazeneca Covid 19 Vaccine Rebel Em Emergency Medicine Blog
 Covid 19 Vaccines November Update Progress Of Clinical Trials Post
Covid 19 Vaccines November Update Progress Of Clinical Trials Post
 Astrazeneca S Covid 19 Vaccine Proves Safe And Effective In Phase Iii Trial
Astrazeneca S Covid 19 Vaccine Proves Safe And Effective In Phase Iii Trial
 Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
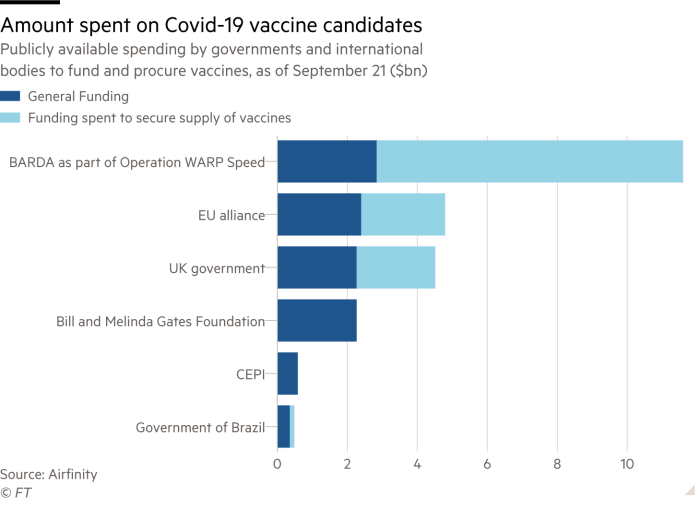 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
Expert Reaction To Preprint From Oxford Looking At Immunogenicity And Efficacy Of The Oxford Astrazeneca Vaccine With A 3 Month Gap Between Doses And Looking At Whether Data Says Anything About Transmission Science
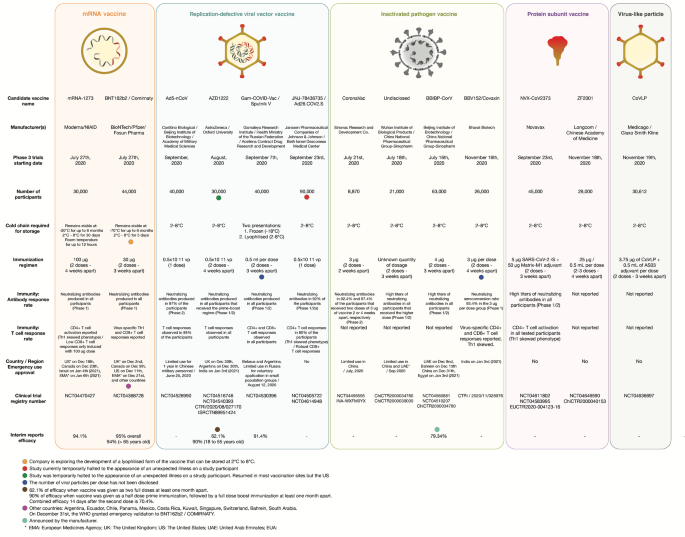 Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
 How Close Is A Coronavirus Vaccine Free To Read Financial Times
How Close Is A Coronavirus Vaccine Free To Read Financial Times
 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
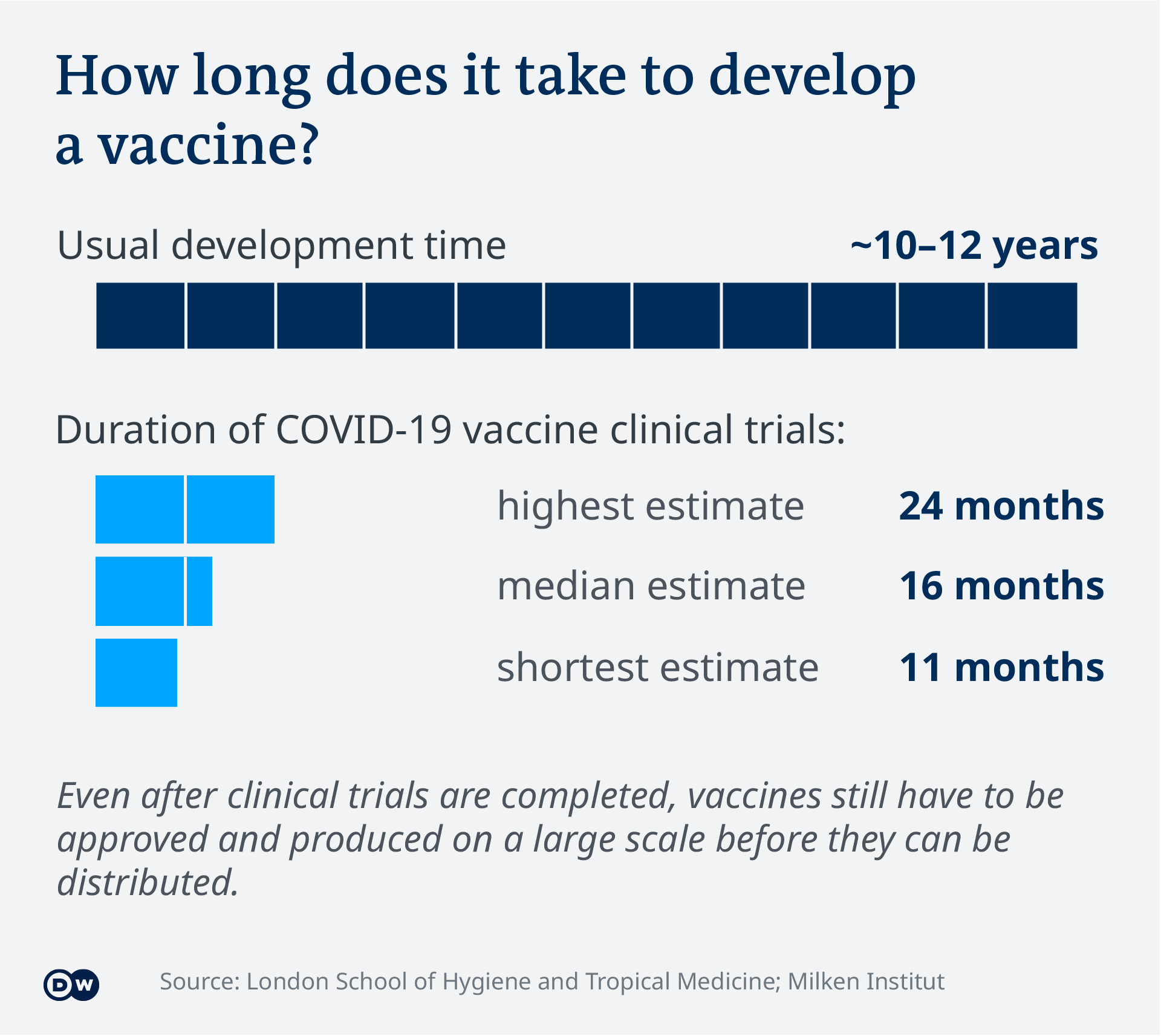 Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 21 04 2021
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Post a Comment for "Astrazeneca Phase 3 Results Uk"