Astrazeneca Phase 3 Covid Trial
Data are first Phase 3 trial results of a coronavirus vaccine to be published in peer-review literature Today University of Oxford and AstraZeneca researchers present a pooled analysis of Phase 3 trials of a vaccine against SARS-CoV-2 across two different dose regimens resulting in an average efficacy of 704. 19012021 An important secondary objective of phase 3 randomized trials will be testing of immune serum from volunteers who were vaccinated and became infected compared to a matched set of vaccinated.
 Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Serum Institute Completes Phase 3 Trial Recruitment For Oxford Astrazeneca Covid 19 Vaccine Hindustan Times
Actual Study Start Date.
Astrazeneca phase 3 covid trial. A multi-site Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. The next stage of. 04022021 AstraZeneca has published the primary analysis results from the Phase III clinical trials of its Covid-19 vaccine in the UK Brazil and South Africa which showed that the vaccine is safe and effective in preventing Covid-19 more than 22 days after the first dose.
AstraZeneca has begun a US. The Brihanmumbai Municipal Corporation BMC has completed the second phase of clinical trials of the Oxford-AstraZeneca Covid-19 vaccine. 31082020 CNN British drugmaker AstraZeneca said Monday it has started Phase 3 trials of its experimental coronavirus vaccine in the United States becoming the.
A Phase III Randomized Double-blind Placebo-controlled Multicenter Study in Adults to Determine the Safety Efficacy and Immunogenicity of AZD1222 a Non-replicating ChAdOx1 Vector Vaccine for the Prevention of COVID-19. 25032021 Positive high-level results from the primary analysis of the phase III trial of AZD1222 in the US have confirmed covid vaccine efficacy Vaccine well-tolerated and. New vaccine efficacy results are reported now in The Lancet.
24032021 AstraZeneca issued updated phase three trial data for its Covid-19 vaccine on Wednesday after facing accuracy questions earlier this week surrounding a preliminary report from its US. The trial will enroll approximately 30000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019. The Biomedical Advanced Research and Development Authority.
An interim analysis of four randomised controlled trials in Brazil South Africa and the UK - The Lancet. With a 76 efficacy rate against symptomatic COVID. 25032021 AstraZeneca said Wednesday that a primary analysis of its Phase 3 trial data confirmed its coronavirus vaccines safety.
Has been put on hold due to a suspected serious. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine AZD1222 against SARS-CoV-2. Phase 3 clinical trial of its COVID-19 vaccine candidate AZD1222.
31 it has begun enrolling up to 30000 people in a phase 3 trial for its experimental COVID-19 vaccine. Researchers at the University of Oxford have begun enrolling subjects in a phase 23 clinical trial of AstraZeneca-partnered COVID-19 vaccine AZD1222. ChAdOx1 nCoV-19 has an acceptable safety profile and has been found to be efficacious against symptomatic COVID-19 in this interim analysis of ongoing clinical trials.
The civic body will soon start third phase trials after. Actual Primary Completion Date. The trial funded by the.
08092020 A large Phase 3 study testing a Covid-19 vaccine being developed by AstraZeneca and the University of Oxford at dozens of sites across the US. Investigators of four randomised controlled trials conducted in the UK South Africa and Brazil report pooled results of an interim analysis of safety and efficacy against COVID-19 of the OxfordAstraZeneca chimpanzee adenovirus vectored vaccine ChAdOx1 nCoV-19 AZD1222 in adults aged 18 years and older. 23112020 When two other pharmaceutical companies Pfizer and Moderna released their efficacy results earlier this month they did include the.
 S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
S Korea Laggard In The Race To Bring Covid 19 Vaccine To Market Pulse By Maeil Business News Korea
 A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
A Timeline Of The Oxford Astrazeneca Covid 19 Vaccine Trials
 Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
Covid 19 Oxford Vaccine Phase 3 Trial Results To Be Out By December Hindustan Times
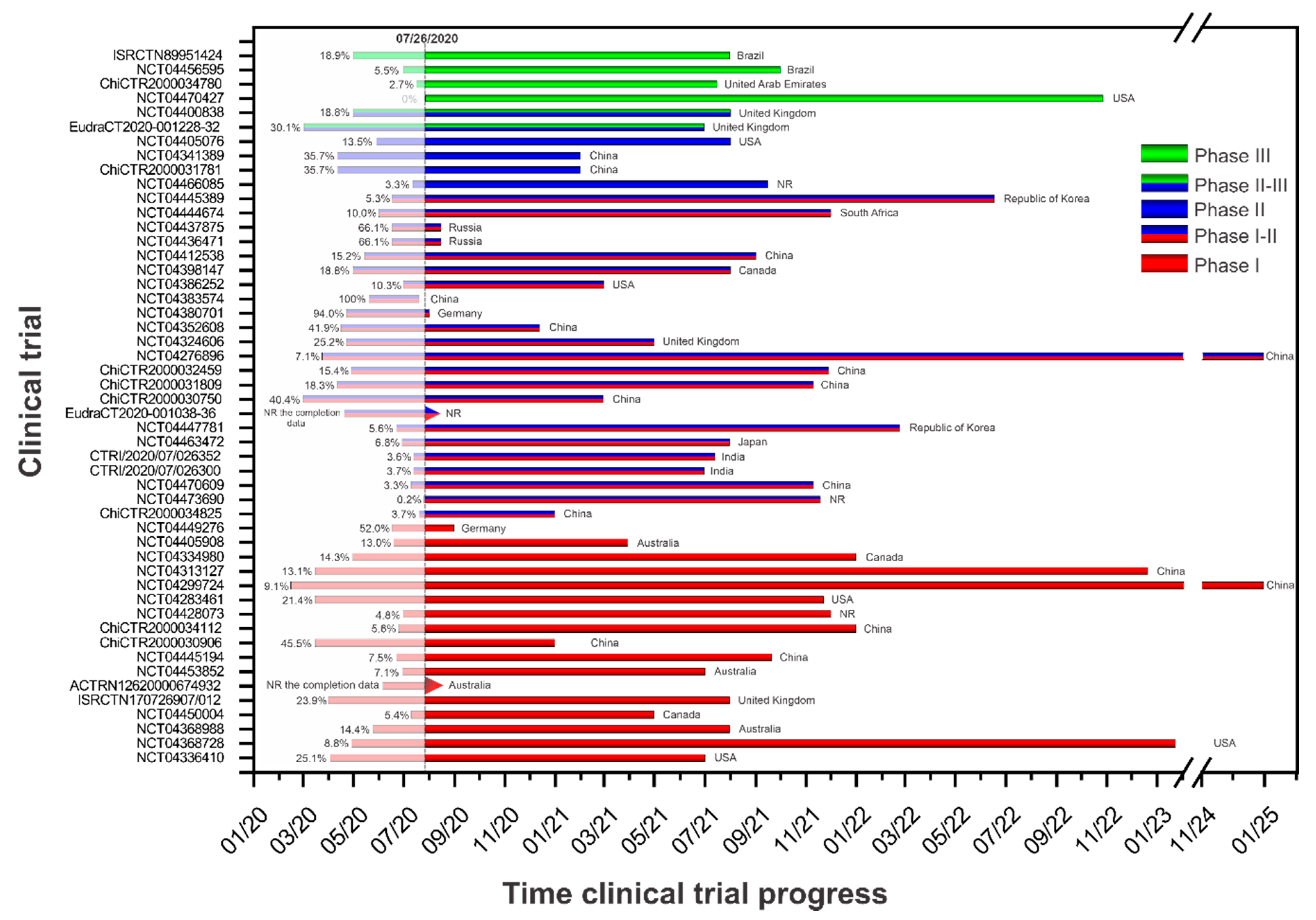 Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
 Astrazeneca S Covid 19 Vaccine Enters Phase 3 Clinical Trial
Astrazeneca S Covid 19 Vaccine Enters Phase 3 Clinical Trial
 Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
Astrazeneca S Quick Covid 19 Vaccine Azd1222 Trial Restart Splits Experts
 Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
Covid 19 Vaccine Update Astrazeneca Admits Manufacturing Error Covaxin Phase 3 Trials To Start In Gujarat Coronavirus Outbreak News
 Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
Covid 19 Update Here S Where We Stand Now In The Race For A Vaccine Tif
 Astrazeneca S Covid 19 Vaccine Candidate Begins Phase 3 Trial Rt
Astrazeneca S Covid 19 Vaccine Candidate Begins Phase 3 Trial Rt
 Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Phase 3 Trial In Us
 Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
Big Relief For Astrazeneca Covid 19 Vaccine Shows 79 Efficacy In Us Phase 3 Trials
 Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3
Coronavirus Vaccine Update Covaxin S Phase 1 Trial To Begin Soon Astrazeneca Sinovac Enter Phase 3


 Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
Covid 19 Vaccine Trial Likely To Conclude By Nov Production May Start In Early 2021 Astrazeneca Hindustan Times
 Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
Astrazeneca Starts Phase 3 Trial For Its Covid 19 Vaccine In Us
 Astrazeneca Covid 19 Vaccine Completes Phase 3 Trial Showing 79 Percent Effectiveness
Astrazeneca Covid 19 Vaccine Completes Phase 3 Trial Showing 79 Percent Effectiveness
 Large Trials For The Oxford Covid 19 Vaccine Begin In The U S
Large Trials For The Oxford Covid 19 Vaccine Begin In The U S
 Oxford Covid 19 Vaccine Shows 70 Efficacy In Phase 2 3 Trials Hindustan Times
Oxford Covid 19 Vaccine Shows 70 Efficacy In Phase 2 3 Trials Hindustan Times
Post a Comment for "Astrazeneca Phase 3 Covid Trial"